
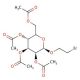
2′-Bromoethyl 2,3,4,6-Tetra-O-acetyl-β-D-glucopyranoside (CAS 16977-78-9)
QUICK LINKS
2′-Bromoethyl 2,3,4,6-Tetra-O-acetyl-β-D-glucopyranoside has emerged as a valuable compound in carbohydrate chemistry and glycobiology research. Its chemical structure features a bromoethyl group attached to a glucopyranoside scaffold with acetyl groups at multiple positions. This chemical has been extensively utilized as a glycosyl donor in glycosylation reactions, particularly in the synthesis of complex oligosaccharides and glycoconjugates. The bromoethyl moiety serves as a leaving group, facilitating the formation of glycosidic bonds with acceptor molecules under appropriate reaction conditions. Researchers have leveraged its reactivity and selectivity to develop efficient strategies for the assembly of diverse glycosidic linkages, enabling the synthesis of structurally-defined carbohydrates for various applications. Additionally, this compound has been employed in the synthesis of glycosylated biomolecules, such as glycopeptides and glycolipids, for studies investigating carbohydrate-protein interactions, cellular recognition processes, and the development of carbohydrate-based probes and sensors. Its utility extends to the construction of carbohydrate microarrays and the preparation of glycosylated surfaces for biosensing applications, highlighting its significance in advancing our understanding of glycobiology and its applications in biotechnology and biomedical research.
2′-Bromoethyl 2,3,4,6-Tetra-O-acetyl-β-D-glucopyranoside (CAS 16977-78-9) References
- Synthesis and properties of new glucocationic surfactants: model structures for marking cationic surfactants with carbohydrates. | Quagliotto, P., et al. 2005. J Org Chem. 70: 9857-66. PMID: 16292816
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2'-Bromoethyl 2,3,4,6-Tetra-O-acetyl-β-D-glucopyranoside, 50 mg | sc-209367 | 50 mg | $360.00 |
