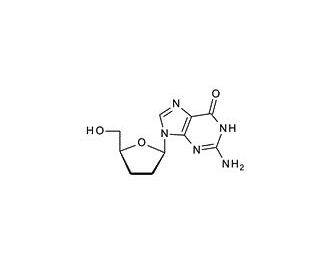
2′,3′-Dideoxyguanosine (CAS 85326-06-3)
QUICK LINKS
2′,3′-Dideoxyguanosine is a nucleoside analog that is of particular interest in the field of molecular biology and genetics. It is incorporated into studies involving the inhibition of nucleic acid synthesis, as the absence of hydroxyl groups at the 2′ and 3′ positions of the sugar moiety prevents further extension of the nucleic acid chain. Researchers utilize 2′,3′-Dideoxyguanosine to understand the mechanisms of DNA replication and to investigate the termination of chain elongation in vitro. This compound is also used in enzymology to study the substrate specificity and kinetics of DNA polymerases and nucleotidyl transferase enzymes. In the area of structural biology, it serves as a tool for crystallography experiments to resolve the three-dimensional structures of nucleic acid-protein complexes. Additionally, 2′,3′-Dideoxyguanosine is relevant in the synthesis of oligonucleotides with specific modifications for use in molecular diagnostics and genetic research.
2′,3′-Dideoxyguanosine (CAS 85326-06-3) References
- Metabolic pathways for the activation of the antiviral agent 2',3'-dideoxyguanosine in human lymphoid cells. | Bondoc, LL., et al. 1992. Mol Pharmacol. 42: 525-30. PMID: 1328848
- Structural analysis of 2',3'-dideoxyinosine, 2',3'-dideoxyadenosine, 2',3'-dideoxyguanosine and 2',3'-dideoxycytidine by 500-MHz 1H-NMR spectroscopy and ab-initio molecular orbital calculations. | Plavec, J., et al. 1992. J Biochem Biophys Methods. 25: 253-72. PMID: 1337354
- Nucleobase transporter-mediated permeation of 2',3'-dideoxyguanosine in human erythrocytes and human T-lymphoblastoid CCRF-CEM cells. | Gati, WP., et al. 1992. J Biol Chem. 267: 22272-6. PMID: 1429579
- Mechanism of inhibition of human immunodeficiency virus type 1 reverse transcriptase and human DNA polymerases alpha, beta, and gamma by the 5'-triphosphates of carbovir, 3'-azido-3'-deoxythymidine, 2',3'-dideoxyguanosine and 3'-deoxythymidine. A novel RNA template for the evaluation of antiretroviral drugs. | Parker, WB., et al. 1991. J Biol Chem. 266: 1754-62. PMID: 1703154
- In vitro inhibition of hepatitis B virus replication by 2',3'-dideoxyguanosine, 2',3'-dideoxyinosine, and 3'-azido-2',3'-dideoxythymidine in 2.2.15 (PR) cells. | Aoki-Sei, S., et al. 1991. J Infect Dis. 164: 843-51. PMID: 1940465
- Liquid chromatographic determination of 2',3'-dideoxyguanosine in human plasma. | Cheung, AP., et al. 1990. J Pharm Biomed Anal. 8: 777-82. PMID: 2100622
- Cellular pharmacology and anti-HIV activity of 2',3'-dideoxyguanosine. | Busso, ME., et al. 1990. AIDS Res Hum Retroviruses. 6: 1139-46. PMID: 2265029
- In Vitro Anti-hepatitis B Virus Activity of 2',3'-Dideoxyguanosine. | Zhang, P., et al. 2018. Virol Sin. 33: 538-544. PMID: 30421112
- Kinetics of the hydrolysis of 2′, 3′-dideoxyguanosine: a potent anti-HIV agent | Van Schepdael, A., Ossembe, N., Jie, L., Herdewijn, P., Roets, E., & Hoogmartens, J. 1991. International journal of pharmaceutics. 73(2): 105-110.
- Complexation of antiretroviral nucleosides 2′, 3′-dideoxyinosine, 2′, 3′-dideoxyadenosine and 2′, 3′-dideoxyguanosine with β-cyclodextrin. A 1 H NMR study | Golankiewicz, B., Perlovich, G., Poznański, J., Sitkowski, J., Zeidler, J., & Zielenkiewicz, W. 1999. Journal of the Chemical Society, Perkin Transactions 2. (11): 2533-2538.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2′,3′-Dideoxyguanosine, 25 mg | sc-283506 | 25 mg | $102.00 | |||
2′,3′-Dideoxyguanosine, 50 mg | sc-283506A | 50 mg | $163.00 |
