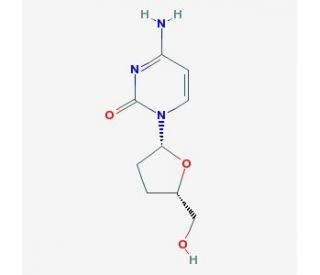
Molecular structure of 2′,3′-Dideoxycytidine, CAS Number: 7481-89-2
2′,3′-Dideoxycytidine (CAS 7481-89-2)
Alternate Names:
ddC
Application:
2′,3′-Dideoxycytidine is an antiviral pyrimidine nucleoside analogue effective against HIV replication
CAS Number:
7481-89-2
Purity:
≥98%
Molecular Weight:
211.22
Molecular Formula:
C9H13N3O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2′,3′-Dideoxycytidine requires activation through phosphorylation by cellular kinases. This activation transforms it into its active form, dideoxycytidine triphosphate (ddCTP). Once activated, ddCTP competes with the natural substrate, deoxycytidine triphosphate (dCTP), for incorporation into the growing DNA chain. This competition leads to the termination of the DNA chain.Various in vivo studies have been conducted to assess the effectiveness of 2′,3′-Dideoxycytidine.
2′,3′-Dideoxycytidine (CAS 7481-89-2) References
- Theoretical mechanistic basis of the toxic effects and efficacy of dideoxycytidine in HIV:AIDS. | Akintonwa, DA. 2001. Med Hypotheses. 57: 249-51. PMID: 11461183
- The molecular basis of inhibition and toxicity of modified cytosine analogues targetting HIV-1 reverse transcriptase. | Anderson, KS. 2001. Antivir Chem Chemother. 12 Suppl 1: 13-7. PMID: 11594679
- Pharmacodynamics of 2',3'-dideoxycytidine: an inhibitor of human immunodeficiency virus. | Broder, S. 1990. Am J Med. 88: 2S-7S. PMID: 1692446
- Zidovudine and other reverse transcriptase inhibitors in the management of human immunodeficiency virus-related disease. | Matthews, SJ., et al. 1991. Pharmacotherapy. 11: 419-48; discussion 448-9. PMID: 1722897
- Treatment of AIDS with combinations of antiretroviral agents. | Merigan, TC. 1991. Am J Med. 90: 8S-17S. PMID: 1850192
- Initial clinical experience with dideoxynucleosides as single agents and in combination therapy. | Yarchoan, R., et al. 1990. Ann N Y Acad Sci. 616: 328-43. PMID: 2078027
- Salvage therapy for zidovudine-intolerant HIV-infected patients with alternating and intermittent regimens of zidovudine and dideoxycytidine. | Bozzette, SA. and Richman, DD. 1990. Am J Med. 88: 24S-26S. PMID: 2159706
- Dideoxycytidine: current clinical experience and future prospects. A summary. | Broder, S. and Yarchoan, R. 1990. Am J Med. 88: 31S-33S. PMID: 2159708
- The antiviral activity of dideoxycytidine. | Jeffries, DJ. 1989. J Antimicrob Chemother. 23 Suppl A: 29-34. PMID: 2541126
- 2',3'-Dideoxycytidine Protects Dopaminergic Neurons in a Mouse Model of Parkinson's Disease. | Niu, J., et al. 2017. Neurochem Res. 42: 2996-3004. PMID: 28631231
- 2',3'-Dideoxycytidine, a DNA Polymerase-β Inhibitor, Reverses Memory Deficits in a Mouse Model of Alzheimer's Disease. | Meng, L., et al. 2019. J Alzheimers Dis. 67: 515-525. PMID: 30584144
- Zalcitabine. An update of its pharmacodynamic and pharmacokinetic properties and clinical efficacy in the management of HIV infection. | Adkins, JC., et al. 1997. Drugs. 53: 1054-80. PMID: 9179531
- 2',3'-Dideoxycytidine cytotoxicity in human macrophages. | Antonelli, A., et al. 1997. Biochim Biophys Acta. 1358: 39-45. PMID: 9296519
Inhibitor of:
HIV-1 RT.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2′,3′-Dideoxycytidine, 100 mg | sc-205579 | 100 mg | $154.00 | |||
2′,3′-Dideoxycytidine, 250 mg | sc-205579A | 250 mg | $333.00 |
