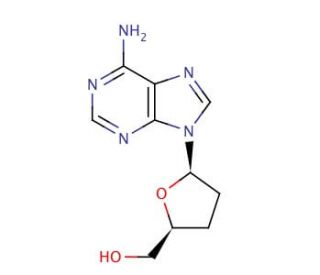
2′,3′-Dideoxyadenosine (CAS 4097-22-7)
See product citations (7)
QUICK LINKS
2′,3′-Dideoxyadenosine (ddA) is a nucleoside analog that plays a crucial role in the study of nucleic acid biochemistry and molecular biology. It features a structural variation where the 2′ and 3′ hydroxyl groups on the ribose sugar are absent, making it an important tool for understanding DNA and RNA synthesis mechanisms. The primary mechanism of action of 2′,3′-Dideoxyadenosine involves its incorporation into DNA or RNA chains during replication or transcription. When ddA is incorporated, it causes premature termination of the nucleic acid chain because it lacks the necessary hydroxyl groups to form a phosphodiester bond with the next nucleotide. This makes ddA an effective chain terminator. In scientific research, ddA is utilized to study the fidelity and mechanics of DNA and RNA polymerases. By incorporating ddA into nucleic acids, researchers can investigate how these enzymes recognize and process substrates, as well as how modifications in nucleoside structure affect these processes. This research provides insights into the enzyme kinetics and dynamics that are fundamental to genetic replication and expression. Additionally, ddA has been employed in molecular biology to map nucleic acid sequences. This usage stems from its ability to terminate DNA synthesis at specific sites, allowing scientists to determine the sequence of nucleotides in DNA through methods such as Sanger sequencing.
2′,3′-Dideoxyadenosine (CAS 4097-22-7) References
- Synthesis of novel 8-substituted carbocyclic analogs of 2',3'-dideoxyadenosine with activity against hepatitis B virus. | Gudmundsson, KS., et al. 2002. Nucleosides Nucleotides Nucleic Acids. 21: 891-901. PMID: 12537029
- Interpretation of the roles of adenylosuccinate lyase and of AMP deaminase in the anti-HIV activity of 2',3'-dideoxyadenosine and 2',3'-dideoxyinosine. | Nair, V. and Sells, TB. 1992. Biochim Biophys Acta. 1119: 201-4. PMID: 1540653
- Pharmacokinetics of 2',3'-dideoxyadenosine in dogs. | Wientjes, MG., et al. 1991. Invest New Drugs. 9: 159-68. PMID: 1908444
- [Conformational capacity of 2',3'-didehydro-2',3'-dideoxyadenosine as a key to understanding its biological activity: results of quantum chemical modelling]. | Ponomar'ova, AH., et al. 2011. Ukr Biokhim Zh (1999). 83: 74-84. PMID: 21851049
- Synthesis and NMR assignment of the two diastereomers of 8,6'-cyclo-2',6'-dideoxyadenosine. | Yueh, H., et al. 2013. Nucleosides Nucleotides Nucleic Acids. 32: 320-32. PMID: 23638925
- Structural and energetic properties of the potential HIV-1 reverse transcriptase inhibitors d4A and d4G: a comprehensive theoretical investigation. | Ponomareva, AG., et al. 2014. J Biomol Struct Dyn. 32: 730-40. PMID: 23947531
- Long-term inhibition of human T-lymphotropic virus type III/lymphadenopathy-associated virus (human immunodeficiency virus) DNA synthesis and RNA expression in T cells protected by 2',3'-dideoxynucleosides in vitro. | Mitsuya, H., et al. 1987. Proc Natl Acad Sci U S A. 84: 2033-7. PMID: 2436223
- Diadenosine diphosphate (Ap₂A) delays neutrophil apoptosis via the adenosine A2A receptor and cAMP/PKA pathway. | Pliyev, BK., et al. 2014. Biochem Cell Biol. 92: 420-4. PMID: 25179165
- Simultaneous determination of 2',3'-dideoxyinosine and the active metabolite, 2',3'-dideoxyadenosine-5'-triphosphate in human peripheral-blood mononuclear cell by HPLC-MS/MS and the application to cell pharmacokinetics. | Lan, X., et al. 2015. J Chromatogr B Analyt Technol Biomed Life Sci. 1002: 337-42. PMID: 26350426
- Synthesis of the 2-chloro analogues of 3'-deoxyadenosine, 2',3'-dideoxyadenosine, and 2',3'-didehydro-2',3'-dideoxyadenosine as potential antiviral agents. | Rosowsky, A., et al. 1989. J Med Chem. 32: 1135-40. PMID: 2785212
- Metabolism and anti-human immunodeficiency virus-1 activity of 2-halo-2',3'-dideoxyadenosine derivatives. | Haertle, T., et al. 1988. J Biol Chem. 263: 5870-5. PMID: 3258602
- Enhancement by 2'-deoxycoformycin of the 5'-phosphorylation and anti-human immunodeficiency virus activity of 2',3'-dideoxyadenosine and 2'-beta-fluoro-2',3'-dideoxyadenosine. | Ahluwalia, GS., et al. 1994. Mol Pharmacol. 46: 1002-8. PMID: 7969062
- Conversion of 2',3'-dideoxyadenosine (ddA) and 2',3'-didehydro-2',3'-dideoxyadenosine (d4A) to their corresponding aryloxyphosphoramidate derivatives markedly potentiates their activity against human immunodeficiency virus and hepatitis B virus. | Balzarini, J., et al. 1997. FEBS Lett. 410: 324-8. PMID: 9237655
- Necrotizing myelopathy in a cat. | Zaki, FA., et al. 1976. J Am Vet Med Assoc. 169: 228-9. PMID: 939717
- Determination of 2'-beta-fluoro-2',3'-dideoxyadenosine, an experimental anti-AIDS drug, in human plasma by high-performance liquid chromatography. | Roth, JS., et al. 1998. J Chromatogr B Biomed Sci Appl. 712: 199-210. PMID: 9698243
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2′,3′-Dideoxyadenosine, 1 mg | sc-202406 | 1 mg | $42.00 | |||
2′,3′-Dideoxyadenosine, 5 mg | sc-202406A | 5 mg | $151.00 | |||
2′,3′-Dideoxyadenosine, 25 mg | sc-202406B | 25 mg | $406.00 |
