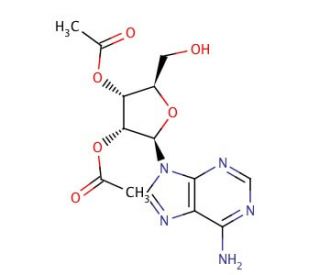
2′,3′-Di-O-acetyladenosine (CAS 29886-19-9)
QUICK LINKS
2′,3′-Di-O-acetyladenosine is a compound utilized in various research fields, including nucleic acid chemistry and enzymology. In studies of nucleic acids, it serves as a modified nucleoside that can be incorporated into oligonucleotides to investigate the effects of acetylation on the structure, stability, and function of RNA and DNA. Enzymologists may employ 2′,3′-Di-O-acetyladenosine to examine the specificity and mechanism of action of nucleoside deacetylases and other enzymes that recognize acetylated substrates. This compound is also a key intermediate in the chemical synthesis of more complex nucleoside derivatives, which are of interest for their potential in studying protein-nucleic acid interactions. Furthermore, it is used in the development of analytical methods for the detection and quantification of acetylated nucleosides. The research involving 2′,3′-Di-O-acetyladenosine enhances the understanding of acetylation′s role in modulating nucleic acid properties and enzyme-substrate interactions.
2′,3′-Di-O-acetyladenosine (CAS 29886-19-9) References
- Mimicking dominant negative inhibition of prion replication through structure-based drug design. | Perrier, V., et al. 2000. Proc Natl Acad Sci U S A. 97: 6073-8. PMID: 10823951
- Synthesis and evaluation of analogues of 5'-([(Z)-4-amino-2-butenyl]methylamino)-5'-deoxyadenosine as inhibitors of tumor cell growth, trypanosomal growth, and HIV-1 infectivity. | Marasco, CJ., et al. 2002. J Med Chem. 45: 5112-22. PMID: 12408722
- Peptidyl-donor substrates for ribosomal peptidyl transferase. Chemical synthesis and biological activity of N-acetyl aminoacyl di- and tri-nucleotides. | Mercer, JF. and Symons, RH. 1972. Eur J Biochem. 28: 38-45. PMID: 5050259
- Synthesis of [35S]thiophosphoryl adenylic acid, utilizing a general procedure for [35S]thiophosphoryl chloride production. | Slama, JT., et al. 1993. Anal Biochem. 209: 143-9. PMID: 8465947
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2′,3′-Di-O-acetyladenosine, 1 g | sc-214079 | 1 g | $71.00 |
