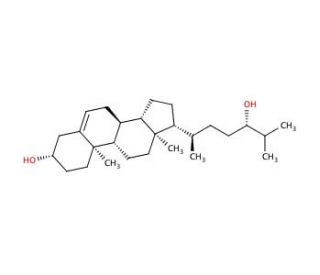
24(S)-Hydroxycholesterol (CAS 474-73-7)
See product citations (1)
QUICK LINKS
24(S)-Hydroxycholesterol plays a pivotal role in the brain′s metabolic processes as a significant cholesterol derivative. Functioning as an endogenous agonist for the liver X receptor (LXR), it engages in regulatory mechanisms that are critical for maintaining cellular cholesterol homeostasis. Its interaction with LXR facilitates the transcription of genes involved in the expulsion of excess cholesterol from cells, underscoring its fundamental role in the intricate balance of cholesterol levels within the brain. Moreover, 24(S)-Hydroxycholesterol exhibits neurotoxic effects on human neuroblastoma cells, indicating its potential impact on neurodegenerative processes by influencing the survival and function of neural cells. This metabolite is also intricately linked with several metabolic pathways, including its involvement in the cerebrotendinous xanthomatosis (CTX) pathway, bile acid biosynthesis, and the pathology of congenital bile acid synthesis defects type II and III. Through its participation in these pathways, 24(S)-Hydroxycholesterol contributes to the complex network of cholesterol metabolism and its derivatives, reflecting its importance not only in neuronal function and health but also in the broader context of lipid homeostasis and metabolic diseases.
24(S)-Hydroxycholesterol (CAS 474-73-7) References
- Profile of cholesterol-related sterols in aged amyloid precursor protein transgenic mouse brain. | Lütjohann, D., et al. 2002. J Lipid Res. 43: 1078-85. PMID: 12091492
- Rediscovery of cerebrosterol. | Björkhem, I. 2007. Lipids. 42: 5-14. PMID: 17393206
- Regulation of alpha- and beta-secretase activity by oxysterols: cerebrosterol stimulates processing of APP via the alpha-secretase pathway. | Famer, D., et al. 2007. Biochem Biophys Res Commun. 359: 46-50. PMID: 17532301
- Complex of amyloid beta peptides with 24-hydroxycholesterol and its effect on hemicholinium-3 sensitive carriers. | Kristofiková, Z., et al. 2008. Neurochem Res. 33: 412-21. PMID: 17717740
- Cholesterol loss during glutamate-mediated excitotoxicity. | Sodero, AO., et al. 2012. EMBO J. 31: 1764-73. PMID: 22343944
- 24S-hydroxycholesterol in plasma: a marker of cholesterol turnover in neurodegenerative diseases. | Leoni, V. and Caccia, C. 2013. Biochimie. 95: 595-612. PMID: 23041502
- Potential diagnostic applications of side chain oxysterols analysis in plasma and cerebrospinal fluid. | Leoni, V. and Caccia, C. 2013. Biochem Pharmacol. 86: 26-36. PMID: 23541982
- The enzyme lecithin-cholesterol acyltransferase esterifies cerebrosterol and limits the toxic effect of this oxysterol on SH-SY5Y cells. | La Marca, V., et al. 2014. J Neurochem. 130: 97-108. PMID: 24620755
- Cholesterol metabolism and homeostasis in the brain. | Zhang, J. and Liu, Q. 2015. Protein Cell. 6: 254-64. PMID: 25682154
- Alterations in cholesterol metabolism as a risk factor for developing Alzheimer's disease: Potential novel targets for treatment. | Loera-Valencia, R., et al. 2019. J Steroid Biochem Mol Biol. 190: 104-114. PMID: 30878503
- Allosteric Modulation of NMDARs Reverses Patients' Autoantibody Effects in Mice. | Radosevic, M., et al. 2022. Neurol Neuroimmunol Neuroinflamm. 9: PMID: 34903638
- Liquid chromatography coupled to tandem mass spectrometry methods for the selective and sensitive determination of 24S-hydroxycholesterol, its sulfate, and/or glucuronide conjugates in plasma. | Brousseau, V., et al. 2022. J Mass Spectrom. 57: e4827. PMID: 35460139
- Sterol metabolism. 28. Biosynthesis and accumulation of cholest-5-ene-3beta, 24-diol (cerebrosterol) in developing rat brain. | Lin, YY. and Smith, LL. 1974. Biochim Biophys Acta. 348: 189-96. PMID: 4847554
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
24(S)-Hydroxycholesterol, 1 mg | sc-471350 | 1 mg | $97.00 | |||
24(S)-Hydroxycholesterol, 5 mg | sc-471350A | 5 mg | $418.00 | |||
24(S)-Hydroxycholesterol, 10 mg | sc-471350B | 10 mg | $774.00 |
