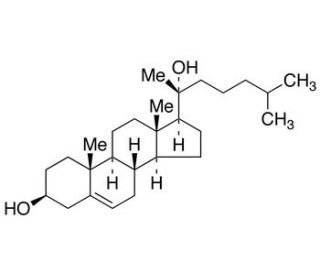
20α-Hydroxy Cholesterol (CAS 516-72-3)
QUICK LINKS
20α-Hydroxycholesterol, a cholesterol derivative, has emerged as a key regulator of cellular cholesterol homeostasis and lipid metabolism. Its mechanism of action primarily involves its role as an endogenous agonist for liver X receptors (LXRs), nuclear receptors that play crucial roles in the transcriptional regulation of genes involved in cholesterol metabolism and transport. Upon binding to LXRs, 20α-hydroxycholesterol induces the expression of genes encoding proteins involved in cholesterol efflux, such as ATP-binding cassette transporters ABCA1 and ABCG1, leading to the removal of excess cholesterol from cells. Additionally, 20α-hydroxycholesterol has been shown to modulate the expression of genes involved in fatty acid synthesis and lipid droplet formation, further influencing lipid metabolism. In scientific research, 20α-hydroxycholesterol has been studied extensively to explain its role in cholesterol homeostasis, lipid metabolism, and the pathogenesis of metabolic disorders such as atherosclerosis and non-alcoholic fatty liver disease (NAFLD). Additionally, 20α-hydroxycholesterol serves as a valuable biomarker for assessing cholesterol metabolism and its dysregulation in various physiological and pathological conditions.
20α-Hydroxy Cholesterol (CAS 516-72-3) References
- Determination of cholesterol oxidation products in milk powder and infant formulas by gas chromatography and mass spectrometry. | Przygonski, K., et al. 2000. Nahrung. 44: 122-5. PMID: 10795581
- Nonionic micellar liquid chromatography coupled to immobilized enzyme reactors. | Tomer, S., et al. 2001. J Chromatogr A. 923: 7-16. PMID: 11510562
- Small Molecule Cocktails Promote Fibroblast-to-Leydig-like Cell Conversion for Hypogonadism Therapy. | Yuan, F., et al. 2023. Pharmaceutics. 15: PMID: 37896216
- Transformation of labeled cholesterol, 20-alpha-hydroxycholesterol, (22R)-22-hydroxycholesterol, and (22R)-20-alpha, 22-dihydroxycholesterol by adrenal acetone-dried preparations from guinea pigs, cattle and man. | Burstein, S., et al. 1970. Steroids. 15: 13-60. PMID: 4905214
- The steatotic and cytotoxic effects of cholesterol oxides in cultured L cells. | Higley, NA. and Taylor, SL. 1984. Food Chem Toxicol. 22: 983-92. PMID: 6542547
- Inhibition of cellular cholesterol efflux by 25-hydroxycholesterol. | Kilsdonk, EP., et al. 1995. J Lipid Res. 36: 505-16. PMID: 7775862
- New developments in congenital lipoid adrenal hyperplasia and steroidogenic acute regulatory protein. | Saenger, P. 1997. Pediatr Clin North Am. 44: 397-421. PMID: 9130927
- The response of the antioxidant defense system in rat hepatocytes challenged with oxysterols is modified by Covi-ox. | Cantwell, H. and Devery, R. 1998. Cell Biol Toxicol. 14: 401-9. PMID: 9879932
- Effects of various cooking and re-heating methods on cholesterol oxidation products of beef loin | Lee, S. O., et al. 2006. Asian-australasian journal of animal sciences. 19(5): 756-762.
- Partial inhibition of cholesterol oxides formation in frozen fish pre‐treated with a plant extract | Lebovics, V., et al. 2009. International journal of food science & technology. 44(2): 342-348.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
20α-Hydroxy Cholesterol, 10 mg | sc-209393 | 10 mg | $300.00 |
