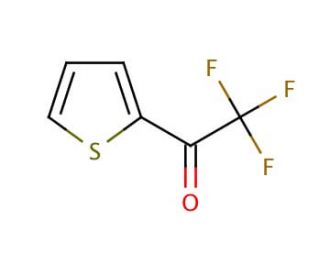
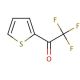
2-(Trifluoroacetyl)thiophene (CAS 651-70-7)
QUICK LINKS
2-(Trifluoroacetyl)thiophene (TFAT) is an organosulfur compound featuring a trifluoroacetyl group bonded to a thiophene ring. 2-(Trifluoroacetyl)thiophene serves as a pivotal intermediate in the production of diverse organic substances, encompassing agrochemicals. It functions as a reagent for generating an array of compounds and materials, making it a versatile foundational component for synthesizing a broad spectrum of molecules and substances. 2-(Trifluoroacetyl)thiophene plays a vital role in numerous scientific research applications, serving as a reagent in the creation of organic compounds. It also plays a pivotal role in crafting polymers, catalysts, and various other materials. 2-(Trifluoroacetyl)thiophene plays a significant role in the synthesis of organic semiconductors, electrochromic materials, and optical materials. In the realm of organic chemistry, 2-(Trifluoroacetyl)thiophene operates as a nucleophile, participating in reactions with electrophiles, such as halides and other organic compounds, to yield novel compounds through nucleophilic substitution. 2-(Trifluoroacetyl)thiophene can act as a leaving group in organic chemistry, facilitating its replacement by alternative compounds.
2-(Trifluoroacetyl)thiophene (CAS 651-70-7) References
- Cloning and overexpression of the Exiguobacterium sp. F42 gene encoding a new short chain dehydrogenase, which catalyzes the stereoselective reduction of ethyl 3-oxo-3-(2-thienyl)propanoate to ethyl (S)-3-hydroxy-3-(2-thienyl)propanoate. | Wada, M., et al. 2004. Biosci Biotechnol Biochem. 68: 1481-8. PMID: 15277752
- Classification structure-activity relationship (CSAR) studies for prediction of genotoxicity of thiophene derivatives. | Du, H., et al. 2008. Toxicol Lett. 177: 10-9. PMID: 18243595
- 3D QSAR of aminophenyl benzamide derivatives as histone deacetylase inhibitors. | Mahipal,., et al. 2010. Med Chem. 6: 277-85. PMID: 20977417
- Synthesis of pentafluorinated β-hydroxy ketones. | Zhang, P. and Wolf, C. 2012. J Org Chem. 77: 8840-4. PMID: 22992005
- Precursor-directed combinatorial biosynthesis of cephalosporin analogue by endolithic actinobacterium Streptomyces sp. AL51 by utilizing thiophene derivative. | Bhattacharjee, K., et al. 2018. 3 Biotech. 8: 31. PMID: 29291144
- Reaction of indoles with aromatic fluoromethyl ketones: an efficient synthesis of trifluoromethyl(indolyl)phenylmethanols using K2CO3/n-Bu4PBr in water. | Pillaiyar, T., et al. 2020. Beilstein J Org Chem. 16: 778-790. PMID: 32395181
- Purification and characterization of fluorinated ketone reductase from Geotrichum candidum NBRC 5767 | Cao, C., Fukae, T., Yamamoto, T., Kanamaru, S., & Matsuda, T. 2013. Biochemical engineering journal. 76: 13-16.
- 2-(Trifluoroacetyl) thiophene as an electrolyte additive for high-voltage lithium-ion batteries using LiCoO2 cathode. | Sun, Y., Huang, J., Xiang, H., Liang, X., Feng, Y., & Yu, Y. 2020. Journal of Materials Science & Technology. 55: 198-202.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-(Trifluoroacetyl)thiophene, 1 g | sc-223275 | 1 g | $34.00 | |||
2-(Trifluoroacetyl)thiophene, 5 g | sc-223275A | 5 g | $117.00 |
