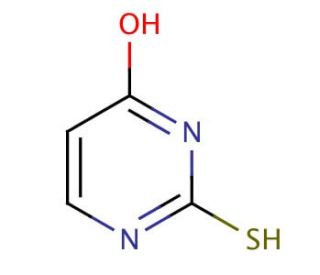
2-Thiouracil (CAS 141-90-2)
QUICK LINKS
2-Thiouracil, also known as 2-thio-4-oxopyrimidine-2-carboxylic acid, is an organic compound containing sulfur that plays a role in various scientific research endeavors. Acting as a structural analog of uracil, a found pyrimidine nucleobase in DNA and RNA, 2-Thiouracil is extensively utilized in biochemical and physiological studies. It holds a significant position in both synthetic and biological processes, proving instrumental in exploring the structures and functions of proteins, enzymes, and other molecules. Notably, 2-Thiouracil boasts diverse applications in scientific research, both in vivo and in vitro. Within living organisms, it serves as a vital tool for studying gene expression regulation, alongside the investigation of protein and enzyme structures and functions. In vitro, it contributes to unraveling the intricacies of nucleic acids and scrutinizing the impacts of various compounds on cellular processes. Although the exact mechanism of action of 2-thiouracil remains elusive, it is postulated to engage with DNA, RNA, proteins, enzymes, and other molecules. Its role as a transcription and translation inhibitor, as well as a gene expression modulator, holds particular interest but warrants further exploration. 2-Thiouracil′s significance as a sulfur-containing compound in scientific research is undeniable, and its applications offer valuable insights into the complex workings of biological systems and molecules.
2-Thiouracil (CAS 141-90-2) References
- Tautomerism in 4-hydroxypyrimidine, S-methyl-2-thiouracil, and 2-thiouracil. | Giuliano, BM., et al. 2010. J Phys Chem A. 114: 12725-30. PMID: 21058636
- 2-Thiouracil deprived of thiocarbonyl function preferentially base pairs with guanine rather than adenine in RNA and DNA duplexes. | Sochacka, E., et al. 2015. Nucleic Acids Res. 43: 2499-512. PMID: 25690900
- Photoelectron spectra of 2-thiouracil, 4-thiouracil, and 2,4-dithiouracil. | Ruckenbauer, M., et al. 2016. J Chem Phys. 144: 074303. PMID: 26896982
- Biomolecules of 2-Thiouracil, 4-Thiouracil and 2,4-Dithiouracil: A DFT Study of the Hydration, Molecular Docking and Effect in DNA:RNAMicrohelixes. | Alcolea Palafox, M., et al. 2019. Int J Mol Sci. 20: PMID: 31311161
- Incorporating 2-Thiouracil into Short Double-Stranded RNA-Binding Peptide Nucleic Acids for Enhanced Recognition of A-U Pairs and for Targeting a MicroRNA Hairpin Precursor. | Ong, AAL., et al. 2019. Biochemistry. 58: 3444-3453. PMID: 31318532
- Cu(II) adsorption on 2-thiouracil-modified Luffa cylindrica biochar fibres from artificial and real samples, and competition reactions with U(VI). | Liatsou, I., et al. 2020. J Hazard Mater. 383: 120950. PMID: 31541960
- Radiation Induced One-Electron Oxidation of 2-Thiouracil in Aqueous Solutions. | Skotnicki, K., et al. 2019. Molecules. 24: PMID: 31810289
- Incorporation of Pseudo-complementary Bases 2,6-Diaminopurine and 2-Thiouracil into Serinol Nucleic Acid (SNA) to Promote SNA/RNA Hybridization. | Kamiya, Y., et al. 2020. Chem Asian J. 15: 1266-1271. PMID: 32020729
- Observation of Enhanced Dissociative Photochemistry in the Non-Native Nucleobase 2-Thiouracil. | Uleanya, KO., et al. 2020. Molecules. 25: PMID: 32664261
- Different Oxidation Pathways of 2-Selenouracil and 2-Thiouracil, Natural Components of Transfer RNA. | Kulik, K., et al. 2020. Int J Mol Sci. 21: PMID: 32825053
- A simple strategy to enhance the luminescence of metal nanoclusters and its application for turn-on detection of 2-thiouracil and hyaluronidase. | Liang, M., et al. 2022. Talanta. 236: 122876. PMID: 34635256
- The Effect of Methylation on the Triplet-State Dynamics of 2-Thiouracil: Time-Resolved Photoelectron Spectroscopy of 2-Thiothymine. | Ullrich, S., et al. 2022. J Phys Chem A. 126: 8211-8217. PMID: 36318646
- Ultrafast Photo-Ion Probing of the Relaxation Dynamics in 2-Thiouracil. | Robinson, MS., et al. 2023. Molecules. 28: PMID: 36903604
- Gold Nanoparticles Functionalized with 2-Thiouracil for Antiproliferative and Photothermal Therapies in Breast Cancer Cells. | Lorenzana-Vázquez, G., et al. 2023. Molecules. 28: PMID: 37298929
- Specific incorporation of 2-thiouracil into biological melanins. | Palumbo, A., et al. 1994. Biochim Biophys Acta. 1200: 271-6. PMID: 8068712
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Thiouracil, 25 g | sc-206492 | 25 g | $60.00 | |||
2-Thiouracil, 100 g | sc-206492A | 100 g | $165.00 |
