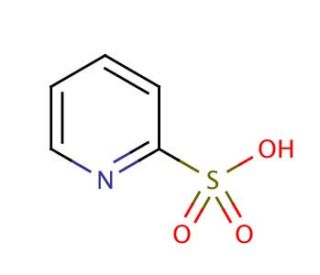
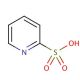
2-Pyridinesulfonic acid (CAS 15103-48-7)
QUICK LINKS
2-Pyridinesulfonic acid is a compound that functions as a catalyst in various chemical reactions, particularly in the synthesis of agrochemicals. It acts as a strong acid, facilitating the formation of new chemical bonds and promoting the rearrangement of molecular structures. 2-Pyridinesulfonic Acid′s mechanism of action involves its ability to donate a proton, which can initiate and accelerate chemical transformations. In experimental applications, 2-Pyridinesulfonic acid is utilized to modify the reactivity of specific functional groups within organic molecules, leading to the production of desired compounds with enhanced properties. Its role in development involves its participation in the creation of novel chemical entities and the optimization of routes for the production of diverse chemical compounds. At the molecular level, 2-Pyridinesulfonic acid interacts with reactant molecules, promoting the activation of specific chemical bonds and enabling the formation of complex molecular architectures.
2-Pyridinesulfonic acid (CAS 15103-48-7) References
- Pyrithiones as antifoulants: environmental fate and loss of toxicity. | Turley, PA., et al. 2005. Biofouling. 21: 31-40. PMID: 16019389
- Aqueous phototransformation of zinc pyrithione Degradation kinetics and byproduct identification by liquid chromatography--atmospheric pressure chemical ionisation mass spectrometry. | Sakkas, VA., et al. 2007. J Chromatogr A. 1144: 175-82. PMID: 17291515
- Well-defined alkylpalladium complexes with pyridine-carboxylate ligands as catalysts for the aerobic oxidation of alcohols. | Melero, C., et al. 2012. Dalton Trans. 41: 14087-100. PMID: 23027588
- Synthesis and characterisation of bismuth(III) aminoarenesulfonate complexes and their powerful bactericidal activity against Helicobacter pylori. | Busse, M., et al. 2013. Chemistry. 19: 5264-75. PMID: 23536213
- S,O-Ligand-Promoted Palladium-Catalyzed C-H Functionalization Reactions of Nondirected Arenes. | Naksomboon, K., et al. 2017. ACS Catal. 7: 6342-6346. PMID: 28966841
- A nuclear permeable Ru(ii)-based photoactivated chemotherapeutic agent towards a series of cancer cells: in vitro and in vivo studies. | Tian, N., et al. 2019. Dalton Trans. 48: 6492-6500. PMID: 30994660
- Method development for on-line species-specific sulfur isotopic analysis by means of capillary electrophoresis/multicollector ICP-mass spectrometry. | Faßbender, S., et al. 2020. Anal Bioanal Chem. 412: 5637-5646. PMID: 32613566
- Microwave-Assisted Synthesis of Organometallic Rhenium (I) Pentylcarbonato Complexes: New Synthon for Carboxylato, Sulfonato and Chlorido Complexes. | Winstead, AJ., et al. 2021. J Organomet Chem. 936: PMID: 33953436
- Development of N-F fluorinating agents and their fluorinations: Historical perspective. | Umemoto, T., et al. 2021. Beilstein J Org Chem. 17: 1752-1813. PMID: 34386101
- Dual ligand approach increases functional group tolerance in the Pd-catalysed C-H arylation of N-heterocyclic pharmaceuticals. | Beckers, I., et al. 2023. Chem Sci. 14: 1176-1183. PMID: 36756333
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Pyridinesulfonic acid, 1 g | sc-225542 | 1 g | $18.00 |
