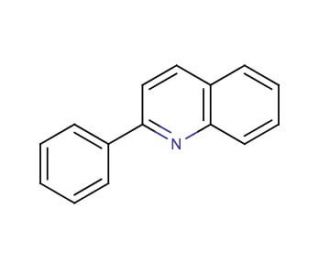
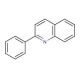
Molecular structure of 2-Phenylquinoline, CAS Number: 612-96-4
2-Phenylquinoline (CAS 612-96-4)
CAS Number:
612-96-4
Molecular Weight:
205.25
Molecular Formula:
C15H11N
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2-Phenylquinoline (2-PQ) is a member of the quinoline family, characterized by its aromatic heterocyclic structure. This compound holds significance in the realm of natural products and actively participates in a variety of biochemical and physiological processes. Scientific research extensively benefits from the applications of 2-Phenylquinoline. It has been utilized in the synthesis of numerous natural products, encompassing terpenes, alkaloids, and flavonoids. 2-Phenylquinoline is recognized for its interaction with enzymes, including cytochrome P450.
2-Phenylquinoline (CAS 612-96-4) References
- Synthesis of 4-alkoxy-2-phenylquinoline derivatives as potent antiplatelet agents. | Ko, TC., et al. 2001. Bioorg Med Chem Lett. 11: 279-82. PMID: 11212091
- Gastroprotective activity of alkaloid extract and 2-phenylquinoline obtained from the bark of Galipea longiflora Krause (Rutaceae). | Zanatta, F., et al. 2009. Chem Biol Interact. 180: 312-7. PMID: 19497430
- Evaluation of antinociceptive effects of Galipea longiflora alkaloid extract and major alkaloid 2-phenylquinoline. | Campos-Buzzi, F., et al. 2010. Methods Find Exp Clin Pharmacol. 32: 707-11. PMID: 21225005
- Descriptor analysis of estrogen receptor β-selective ligands using 2-phenylquinoline, tetrahydrofluorenone and 3-hydroxy 6H-benzo[c]chromen-6-one scaffolds. | Balaji, ., et al. 2011. J Enzyme Inhib Med Chem. 26: 831-42. PMID: 21438712
- Highly efficient electrochemiluminescence from iridium(III) complexes with 2-phenylquinoline ligand. | Zhou, Y., et al. 2015. Dalton Trans. 44: 1858-65. PMID: 25482203
- The 'racemic approach' in the evaluation of the enantiomeric NorA efflux pump inhibition activity of 2-phenylquinoline derivatives. | Carotti, A., et al. 2016. J Pharm Biomed Anal. 129: 182-189. PMID: 27429367
- Boosting Effect of 2-Phenylquinoline Efflux Inhibitors in Combination with Macrolides against Mycobacterium smegmatis and Mycobacterium avium. | Machado, D., et al. 2015. ACS Infect Dis. 1: 593-603. PMID: 27623057
- Gastroprotective and anti-secretory mechanisms of 2-phenylquinoline, an alkaloid isolated from Galipea longiflora. | Breviglieri, E., et al. 2017. Phytomedicine. 25: 61-70. PMID: 28190472
- Mode of action of the 2-phenylquinoline efflux inhibitor PQQ4R against Escherichia coli. | Machado, D., et al. 2017. PeerJ. 5: e3168. PMID: 28516003
- 2-Phenylquinoline S. aureus NorA Efflux Pump Inhibitors: Evaluation of the Importance of Methoxy Group Introduction. | Felicetti, T., et al. 2018. J Med Chem. 61: 7827-7848. PMID: 30067360
- Ir-Ho bimetallic complex-mediated low-dose radiotherapy/radiodynamic therapy in vivo. | Wang, C., et al. 2020. Chem Commun (Camb). 56: 6193-6196. PMID: 32432578
- Unsymmetric Heteroleptic Ir(III) Complexes with 2-Phenylquinoline and Coumarin-Based Ligand Isomers for Tuning Character of Triplet Excited States and Achieving High Electroluminescent Efficiencies. | Feng, Z., et al. 2020. Inorg Chem. 59: 12362-12374. PMID: 32799532
- Discrepancy between Proline and Homoproline in Chiral Recognition and Diastereomeric Photoreactivity with Iridium(III) Complexes. | Xiong, MF., et al. 2021. Inorg Chem. 60: 5423-5431. PMID: 33818063
- The interaction of substituted 2-phenylquinoline intercalators with poly(A).poly(U): classical and threading intercalation modes with RNA. | Zhao, M., et al. 1994. Biopolymers. 34: 61-73. PMID: 7509202
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Phenylquinoline, 1 g | sc-230643 | 1 g | $47.00 |
