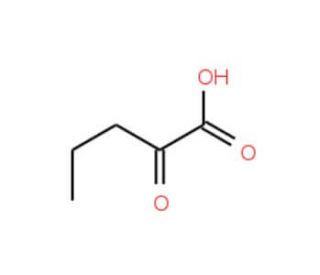
Molecular structure of 2-Oxovaleric acid, CAS Number: 1821-02-9
2-Oxovaleric acid (CAS 1821-02-9)
Alternate Names:
α-Ketovaleric acid
CAS Number:
1821-02-9
Purity:
≥95%
Molecular Weight:
116.12
Molecular Formula:
C5H8O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2-Oxopentanoic acid, also referred to as 2-oxo-pentanoic acid, is an alpha-keto acid, playing a vital role as an intermediate in the metabolism of both fatty acids and amino acids. Its significance extends beyond metabolism, finding applications in the synthesis of various organic compounds. The conversion of 2-oxopentanoic acid into acetyl-CoA, facilitated by the enzyme acyl-CoA synthetase, serves as a key step in the metabolic breakdown of fatty acids and amino acids. Acetyl-CoA then participates in the citric acid cycle, yielding cellular energy for various cellular processes.
2-Oxovaleric acid (CAS 1821-02-9) References
- The mechanism of the formation of higher alcohols from amino acids by Saccharomyces cerevisiae. | SENTHESHANUGANATHAN, S. 1960. Biochem J. 74: 568-76. PMID: 13856344
- Inhibition of malate-aspartate shuttle by the antitumor drug L-glutamic acid gamma-monohydroxamate in L1210 leukemia cells. | Thomasset, N., et al. 1992. Int J Cancer. 51: 329-32. PMID: 1568800
- Modified branched-chain amino acid pathways give rise to acyl acids of sucrose esters exuded from tobacco leaf trichomes. | Kandra, G., et al. 1990. Eur J Biochem. 188: 385-91. PMID: 2318213
- Intermediate role of α-keto acids in the formation of Strecker aldehydes. | Hidalgo, FJ., et al. 2013. Food Chem. 141: 1140-6. PMID: 23790896
- Metal-Dependent Function of a Mammalian Acireductone Dioxygenase. | Deshpande, AR., et al. 2016. Biochemistry. 55: 1398-407. PMID: 26858196
- Plasma metabonomics study of first-Episode schizophrenia treated with olanzapine in female patients. | Qiao, Y., et al. 2016. Neurosci Lett. 617: 270-6. PMID: 26924724
- Essential role of amino acid position 71 in substrate preference by meso-diaminopimelate dehydrogenase from Symbiobacterium thermophilum IAM14863. | Zhang, Y., et al. 2018. Enzyme Microb Technol. 111: 57-62. PMID: 29421037
- Characterization of Chemosensory Responses on the Labellum of the Malaria Vector Mosquito, Anopheles coluzzii. | Saveer, AM., et al. 2018. Sci Rep. 8: 5656. PMID: 29618749
- Ir76b is a Co-receptor for Amine Responses in Drosophila Olfactory Neurons. | Vulpe, A. and Menuz, K. 2021. Front Cell Neurosci. 15: 759238. PMID: 34867202
- NMR as powerful technology for noninvasively monitoring cell health and expansion during bioprocessing. | Benevelli, F., et al. 2022. Biotechnol Bioeng. 119: 3497-3508. PMID: 36000349
- Infrared Reflection-Absorption Spectroscopy of α-Keto Acids at the Air-Water Interface: Effects of Chain Length and Headgroup on Environmentally Relevant Surfactant Films. | Deal, AM., et al. 2023. J Phys Chem A. 127: 4137-4151. PMID: 37103984
- Influence of pH on the allosteric properties of lactate dehydrogenase activity of Phycomyces blakesleeanus. | De Arriaga, D., et al. 1982. Biochem J. 203: 393-400. PMID: 7115294
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Oxovaleric acid, 1 ml | sc-230617 | 1 ml | $183.00 | |||
2-Oxovaleric acid, 5 ml | sc-230617A | 5 ml | $501.00 |
