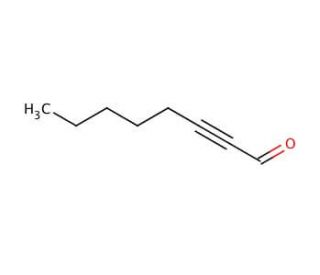
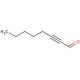
2-Octynal (CAS 1846-68-0)
QUICK LINKS
2-Octynal, a chemical compound classified as an aldehyde, possesses notable characteristics. It exists as a colorless liquid with a potent odor and finds extensive application in diverse scientific research endeavors. The versatility of 2-Octynal makes it highly valuable in numerous scientific research applications. It is employed for organic compound synthesis, investigating enzyme-catalyzed reactions, and exploring protein-ligand interactions. Through studies, 2-Octynal has been identified as an inhibitor of several enzymes, including acetylcholinesterase, butyrylcholinesterase, and monoamine oxidase. Additionally, it interacts with various proteins such as albumin and hemoglobin. Notably, 2-Octynal forms covalent adducts with cysteine residues in proteins, thereby inducing alterations in protein structure and function.
2-Octynal (CAS 1846-68-0) References
- Lipase-mediated resolution of 4-TMS-3-butyn-2-ol and use of the mesylate derivatives as a precursor to a highly stereoselective chiral allenylindium reagent. | Marshall, JA., et al. 2001. Org Lett. 3: 3369-72. PMID: 11594836
- Interspecies quantitative structure-activity relationship model for aldehydes: aquatic toxicity. | Dimitrov, S., et al. 2004. Environ Toxicol Chem. 23: 463-70. PMID: 14982395
- Enantioselective syntheses of (R)- and (S)-argentilactone and their cytotoxic activities against cancer cell lines. | de Fatima, A., et al. 2004. Bioorg Med Chem. 12: 5437-42. PMID: 15388170
- An efficient synthesis of epoxydiynes and a key fragment of neocarzinostatin chromophore. | Baker, JR., et al. 2007. Org Lett. 9: 45-8. PMID: 17192081
- Synthesis of a fluorous ligand and its application for asymmetric addition of dimethylzinc to aldehydes. | Sokeirik, YS., et al. 2007. Org Lett. 9: 1927-9. PMID: 17425326
- Thiol reactivity and its impact on the ciliate toxicity of α,β-unsaturated aldehydes, ketones, and esters. | Böhme, A., et al. 2010. Chem Res Toxicol. 23: 1905-12. PMID: 20923215
- Stereochemistry of hydrogen removal during oxygenation of linoleic acid by singlet oxygen and synthesis of 11(S)-deuterium-labeled linoleic acid. | Hamberg, M. 2011. Lipids. 46: 201-6. PMID: 21161604
- Copper(I)-catalyzed enantioselective synthesis of α-chiral linear or carbocyclic (E)-(γ-alkoxyallyl)boronates. | Yamamoto, E., et al. 2014. J Am Chem Soc. 136: 16515-21. PMID: 25329863
- Chemoavailability of Organic Electrophiles: Impact of Hydrophobicity and Reactivity on Their Aquatic Excess Toxicity. | Böhme, A., et al. 2016. Chem Res Toxicol. 29: 952-62. PMID: 27096880
- Selective Catalytic Hydrogenations of Nitriles, Ketones, and Aldehydes by Well-Defined Manganese Pincer Complexes. | Elangovan, S., et al. 2016. J Am Chem Soc. 138: 8809-14. PMID: 27219853
- Iridium versus Iridium: Nanocluster and Monometallic Catalysts Carrying the Same Ligand Behave Differently. | Cano, I., et al. 2017. Chemistry. 23: 1444-1450. PMID: 27873441
- In situ generation of less accessible Boc-imines from aldehydes: construction of a quaternary carbon by the Mannich reaction or unprecedented aldol reaction. | Kano, T., et al. 2017. Org Biomol Chem. 15: 4527-4530. PMID: 28513717
- Studies on the metabolism of unsaturated fatty acids. XVI. Purification and general properties of 2,4-dienoyl-CoA reductase from Candida lipolytica. | Mizugaki, M., et al. 1985. J Biochem. 97: 837-43. PMID: 4019437
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Octynal, 5 g | sc-225521 | 5 g | $224.00 |
