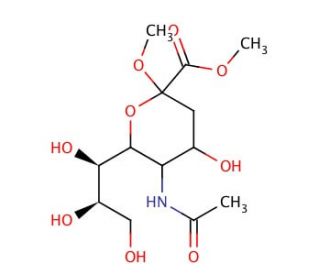
2-O-Methyl-β-D-N-acetylneuraminic Acid, Methyl Ester (CAS 6730-43-4)
QUICK LINKS
2-O-Methyl-β-D-N-acetylneuraminic acid, methyl ester, is a chemical compound with significant implications in glycobiology and immunology research. Its mechanism of action revolves around its role as a sialic acid derivative, a class of molecules crucial for various biological processes, including cell-cell interactions, signal transduction, and immune response modulation. This compound serves as a valuable tool in studying sialic acid biology, particularly in elucidating its role in cellular recognition and immune evasion mechanisms. Researchers utilize 2-O-Methyl-β-D-N-acetylneuraminic acid, methyl ester, in experiments aimed at investigating the binding specificity of sialic acid-binding proteins, such as lectins and viral hemagglutinins. Moreover, it is employed in glycomics studies to analyze the sialylation patterns of glycoproteins and glycolipids, providing insights into their functional roles in health and disease. Additionally, this compound finds application in the development of glycan-based vaccines, where understanding sialic acid interactions is crucial for designing targeted interventions against pathogens and cancer cells. Its versatile utility in glycobiology research underscores its significance as a molecular probe for elucidating complex biological processes involving sialic acids.
2-O-Methyl-β-D-N-acetylneuraminic Acid, Methyl Ester (CAS 6730-43-4) References
- Carbohydrate materials bearing neuraminidase-resistant C-glycosides of sialic acid strongly inhibit the in vitro infectivity of influenza virus. | Nagy, JO., et al. 1992. J Med Chem. 35: 4501-2. PMID: 1447751
- Lectins as cell recognition molecules. | Sharon, N. and Lis, H. 1989. Science. 246: 227-34. PMID: 2552581
- Hemagglutinins from two influenza virus variants bind to sialic acid derivatives with millimolar dissociation constants: a 500-MHz proton nuclear magnetic resonance study. | Sauter, NK., et al. 1989. Biochemistry. 28: 8388-96. PMID: 2605190
- Binding determinants of the sialic acid-specific lectin from the slug Limax flavus. | Knibbs, RN., et al. 1993. J Biol Chem. 268: 18524-31. PMID: 7689555
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-O-Methyl-β-D-N-acetylneuraminic Acid, Methyl Ester, 100 mg | sc-220760 | 100 mg | $300.00 |
