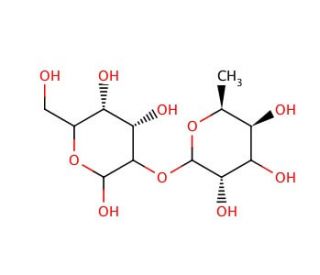
2-O-α-L-Fucopyranosyl-D-galactose (CAS 24656-24-4)
QUICK LINKS
2-O-α-L-Fucopyranosyl-D-galactose is a compound of interest in scientific research, particularly in the field of glycobiology. This chemical plays a crucial role as a glycan component in various biological processes, including cell-cell recognition, cell adhesion, and immune response modulation. One notable research application involves its involvement in the biosynthesis of glycoconjugates and glycoproteins, where it serves as a key intermediate in the construction of complex oligosaccharide structures. Researchers utilize this compound as a substrate in enzymatic assays to investigate the specificity and catalytic mechanisms of glycosyltransferases and glycosidases involved in glycan biosynthesis and modification pathways. Furthermore, 2-O-α-L-Fucopyranosyl-D-galactose has been utilized in the synthesis of glycan-based probes and glycomimetics for studying carbohydrate-protein interactions and elucidating the roles of glycans in various biological processes. Its ability to mimic natural glycan structures makes it valuable in designing and developing glycan-based vaccines, and diagnostics. Additionally, this compound serves as a useful tool in glycoengineering strategies aimed at modulating cellular functions and signaling pathways for research purposes. Overall, 2-O-α-L-Fucopyranosyl-D-galactose contributes significantly to advancing our understanding of glycobiology and has promising applications in biomedical research and biotechnology.
2-O-α-L-Fucopyranosyl-D-galactose (CAS 24656-24-4) References
- Crystal structure of fucose-specific lectin from Aleuria aurantia binding ligands at three of its five sugar recognition sites. | Fujihashi, M., et al. 2003. Biochemistry. 42: 11093-9. PMID: 14503859
- Demonstration by monoclonal antibodies that carbohydrate structures of glycoproteins and glycolipids are onco-developmental antigens. | Feizi, T. Nature. 314: 53-7. PMID: 2579340
- Glycosidases of aspergillus niger. II. Purification and general properties of 1,2-alpha-L-fucosidase. | Bahl, OP. 1970. J Biol Chem. 245: 299-304. PMID: 5460888
- Crystal state and solution conformation of the B blood group trisaccharide alpha-L-Fucp-(1-->2)-[alpha-D-Galp]-(1-->3)]-beta-D-Galp-OCH3. | Otter, A., et al. 1999. Eur J Biochem. 259: 295-303. PMID: 9914506
- Synthesis of benzyl 6-O-benzoyl-3,4-O-isopropylidene-β-D-galactopyranoside and of 2-O-α-L-fucopyranosyl-D-galactose | A. Levy, H.M. Flowers, N. Sharon. 1967. Carbohydrate Research. 4: 305-311.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-O-α-L-Fucopyranosyl-D-galactose, 5 mg | sc-220755 | 5 mg | $390.00 |
