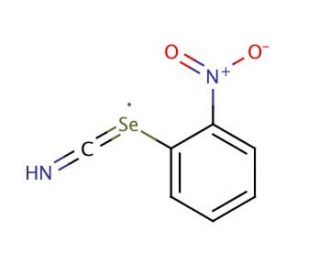
2-Nitrophenyl selenocyanate (CAS 51694-22-5)
QUICK LINKS
2-Nitrophenyl selenocyanate is an organoselenium compound that has gained interest within the field of organic chemistry for its use in the introduction of selenium-containing groups into organic molecules. Its reactivity is particularly useful in the synthesis of selenoamides and selenoureas, which are explored for their unique chemical and physical properties. The nitrophenyl moiety acts as a good leaving group, which is beneficial in nucleophilic substitution reactions, making 2-Nitrophenyl selenocyanate a versatile reagent for the formation of carbon-selenium bonds. Studies involving 2-nitrophenyl selenocyanate focus on the synthesis of biologically relevant selenium compounds, contributing to the understanding of selenium′s role in chemical biology. 2-Nitrophenyl selenocyanate is in the preparation of selenium-based catalysts, which are of growing interest in the development of green chemistry applications due to their ability to facilitate reactions under milder conditions and with higher selectivity compared to their sulfur analogs.
2-Nitrophenyl selenocyanate (CAS 51694-22-5) References
- Formal total synthesis of (+)-trehazolin. Application of an asymmetric aldol-olefin metathesis approach to the synthesis of functionalized cyclopentenes. | Crimmins, MT. and Tabet, EA. 2001. J Org Chem. 66: 4012-8. PMID: 11375027
- Synthesis of labeled 1-amino-2-methylenecyclopropane-1-carboxylic acid, an inactivator of 1-aminocyclopropane-1-carboxylate deaminase. | Zhao, Z. and Liu, HW. 2002. J Org Chem. 67: 2509-14. PMID: 11950295
- trans-6-Aminocyclohept-3-enols, a new designed polyfunctionalized chiral building block for the asymmetric synthesis of 2-substituted-4-hydroxypiperidines. | Celestini, P., et al. 2002. Org Lett. 4: 1367-70. PMID: 11950364
- Synthesis of the bicyclo[7.3.0]dodecatrienediyne core of the C-1027 chromophore. | Inoue, M., et al. 2004. Org Lett. 6: 3833-6. PMID: 15469361
- Synthesis of an optically pure synthetic intermediate of aloperine from a yeast-reductive product. | Yamauchi, S. and Omi, Y. 2005. Biosci Biotechnol Biochem. 69: 1589-94. PMID: 16116290
- 9-{[3-fluoro-2-(hydroxymethyl)cyclopropylidene]methyl}adenines and -guanines. Synthesis and antiviral activity of all stereoisomers1. | Zhou, S., et al. 2006. J Med Chem. 49: 6120-8. PMID: 17004726
- Critical importance of the nine-membered F ring of ciguatoxin for potent bioactivity: total synthesis and biological evaluation of F-ring-modified analogues. | Inoue, M., et al. 2008. Angew Chem Int Ed Engl. 47: 8611-4. PMID: 18846538
- Breaking and making of olefins simultaneously using ozonolysis: application to the synthesis of useful building blocks and macrocyclic core of solomonamides. | Kashinath, K., et al. 2015. Org Lett. 17: 2090-3. PMID: 25870910
- Synthesis of 3,4-Fused Tricyclic Indoles Using 3-Alkylidene Indolines as Versatile Precursors. | Nemoto, T. 2019. Chem Rec. 19: 320-332. PMID: 29931736
- Expedient Total Syntheses of Pladienolide-Derived Spliceosome Modulators. | Rhoades, D., et al. 2021. J Am Chem Soc. 143: 4915-4920. PMID: 33755462
- Redox-Neutral Selenium-Catalysed Isomerisation of para-Hydroxamic Acids into para-Aminophenols. | Chuang, HY., et al. 2021. Angew Chem Int Ed Engl. 60: 13778-13782. PMID: 33760338
- Phosphine-Catalyzed Aryne Oligomerization: Direct Access to α,ω-Bisfunctionalized Oligo(ortho-arylenes). | Bürger, M., et al. 2021. J Am Chem Soc. 143: 16796-16803. PMID: 34585921
- Synthesis of 4 alpha-(2-propenyl)-5,6-secocholestan-3 alpha-ol, a novel B-ring seco analog of the hypocholesterolemic agent 4 alpha-(2-propenyl)-5 alpha-cholestan-3 alpha-ol. | Lin, HS., et al. 1998. Steroids. 63: 202-7. PMID: 9589554
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Nitrophenyl selenocyanate, 1 g | sc-238176 | 1 g | $105.00 |
