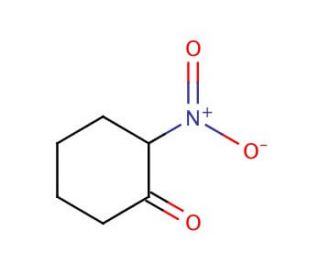
2-Nitrocyclohexanone (CAS 4883-67-4)
QUICK LINKS
2-Nitrocyclohexanone is a chemical compound that functions as a reactant in organic synthesis. It acts as a precursor in the preparation of various organic compounds, including intermediates and fine chemicals. The mechanism of action of 2-Nitrocyclohexanone involves its participation in nucleophilic addition reactions, leading to the formation of new carbon-carbon or carbon-heteroatom bonds. 2-Nitrocyclohexanone undergoes chemical transformations, such as reduction or substitution, to yield products with diverse chemical functionalities. In the experimental, 2-Nitrocyclohexanone serves as a building block for the synthesis of complex molecules, enabling the creation of novel chemical structures with potential applications in various fields. Its reactivity and ability to undergo selective transformations make it a versatile component in the development of new chemical entities.
2-Nitrocyclohexanone (CAS 4883-67-4) References
- Determination of the polarities of some ionic liquids using 2-nitrocyclohexanone as the probe. | Angelini, G., et al. 2005. J Org Chem. 70: 8193-6. PMID: 16277347
- Ionization and tautomerization of 2-nitrocyclohexanone in aqueous solution. | Angelini, G., et al. 2007. J Org Chem. 72: 4039-47. PMID: 17451273
- Solvent effects on the rate of the keto-enol interconversion of 2-nitrocyclohexanone. | Siani, G., et al. 2008. Org Biomol Chem. 6: 4236-41. PMID: 18972056
- The base-catalyzed keto-enol interconversion of 2-nitrocyclohexanone in ionic liquids. | Angelini, G., et al. 2009. J Org Chem. 74: 6572-6. PMID: 19637884
- Reductive cross-coupling of nonaromatic, heterocyclic bromides with aryl and heteroaryl bromides. | Molander, GA., et al. 2014. J Org Chem. 79: 5771-80. PMID: 24892751
- An organocatalytic cascade reaction of 2-nitrocyclohexanone and α,β-unsaturated aldehydes with unusual regioselectivity. | Xuan, YN., et al. 2014. Chem Commun (Camb). 50: 10471-3. PMID: 25068210
- Transition-metal-free α-arylation of nitroketones with diaryliodonium salts for the synthesis of tertiary α-aryl, α-nitro ketones. | An, Y., et al. 2018. Chem Commun (Camb). 55: 119-122. PMID: 30516178
- Bis-enolates with Extended π-Conjugation Are Powerful Nucleophiles: A Study of Their Alkylation Reactions with Very Hindered C-Electrophiles. | Castroagudín, M., et al. 2019. J Org Chem. 84: 15805-15816. PMID: 31778066
- Characterization of new highly selective pyrazolo[4,3-d]pyrimidine inhibitor of CDK7. | Kovalová, M., et al. 2023. Biomed Pharmacother. 161: 114492. PMID: 36931035
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Nitrocyclohexanone, 1 g | sc-230597 | 1 g | $102.00 |
