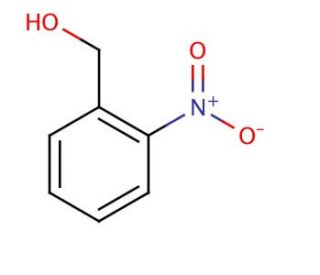
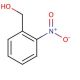
2-Nitrobenzyl alcohol (CAS 612-25-9)
QUICK LINKS
2-Nitrobenzyl alcohol functions as a protecting group in organic synthesis. It is used to temporarily mask the hydroxyl group of alcohols and phenols, allowing for selective reactions to occur at other functional groups. The mechanism of action involves the formation of a covalent bond between the hydroxyl group of the alcohol or phenol and the nitrobenzyl group, effectively blocking its reactivity. This protection can be reversed under specific conditions, such as exposure to ultraviolet light, allowing for the recovery of the original hydroxyl functionality. In this way, 2-Nitrobenzyl alcohol is useful for controlling the reactivity of hydroxyl-containing compounds in various synthesis pathways, enabling the synthesis of complex molecules with precise structural modifications.
2-Nitrobenzyl alcohol (CAS 612-25-9) References
- Photochemical reaction mechanisms of 2-nitrobenzyl compounds: 2-nitrobenzyl alcohols form 2-nitroso hydrates by dual proton transfer. | Gaplovsky, M., et al. 2005. Photochem Photobiol Sci. 4: 33-42. PMID: 15616689
- Near-infrared light-triggered micelles for fast controlled drug release in deep tissue. | Cao, J., et al. 2013. Biomaterials. 34: 6272-83. PMID: 23721796
- Kinetics of conjugation and oxidation of nitrobenzyl alcohols by rat hepatic enzymes. | Rickert, DE., et al. 1985. Biochem Pharmacol. 34: 4163-8. PMID: 2933044
- Phototriggered labeling and crosslinking by 2-nitrobenzyl alcohol derivatives with amine selectivity. | Wang, C., et al. 2020. Chem Commun (Camb). 56: 2264-2267. PMID: 31984385
- Intramolecular redox cyclization reaction access to cinnolines from 2-nitrobenzyl alcohol and benzylamine via intermediate 2-nitrosobenzaldehyde. | Sa, Y., et al. 2022. RSC Adv. 12: 33260-33263. PMID: 36425163
- Metabolism of nitrotoluenes by freshly isolated Fischer 344 rat hepatocytes. | deBethizy, JD. and Rickert, DE. 1984. Drug Metab Dispos. 12: 45-50. PMID: 6141911
- Light activatable antibodies: models for remotely activatable proteins. | Self, CH. and Thompson, S. 1996. Nat Med. 2: 817-20. PMID: 8673931
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Nitrobenzyl alcohol, 25 g | sc-238172 | 25 g | $47.00 |
