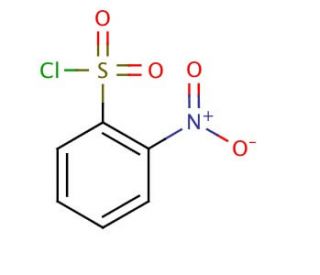
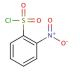
2-Nitrobenzenesulfonyl chloride (CAS 1694-92-4)
QUICK LINKS
2-Nitrobenzenesulfonyl chloride is a chemical compound that functions as an acylating agent in organic synthesis. It is used to introduce the sulfonyl chloride functional group into various organic molecules, allowing for the modification of their chemical properties. The mode of action of 2-Nitrobenzenesulfonyl chloride involves the reaction of the sulfonyl chloride group with nucleophilic functional groups, such as amines or alcohols, to form covalent bonds. This reaction results in the formation of sulfonamide or sulfonate ester derivatives, which can be further manipulated in organic synthesis. Mode of action at the molecular level involves the transfer of the sulfonyl chloride group to the nucleophilic substrate, leading to the formation of a new chemical bond. This allows for the selective modification of specific functional groups within organic molecules, enabling the synthesis of diverse chemical compounds for development purposes.
2-Nitrobenzenesulfonyl chloride (CAS 1694-92-4) References
- Solid-phase synthesis and chemical properties of 2-(2-amino/hydroxyethyl)-1-aryl-3,4-dihydropyrazino[1, 2-b]indazol-2-iums. | Kocí, J. and Krchnák, V. 2010. J Comb Chem. 12: 168-75. PMID: 19961214
- N-Pyrrylarylsulfones with High Therapeutic Potential. | Famiglini, V., et al. 2017. Molecules. 22: PMID: 28282943
- Recent Reports of Solid-Phase Cyclohexapeptide Synthesis and Applications. | Prior, AM., et al. 2018. Molecules. 23: PMID: 29912160
- Enhancing the ligand efficiency of anti-HIV compounds targeting frameshift-stimulating RNA. | Anokhina, VS., et al. 2019. Bioorg Med Chem. 27: 2972-2977. PMID: 31101492
- Sultam-Based Hetero[5]helicene: Synthesis, Structure, and Crystallization-Induced Emission Enhancement. | Virk, TS., et al. 2016. ACS Omega. 1: 1336-1342. PMID: 31457199
- 1,2,4-Triazine Sulfonamides: Synthesis by Sulfenamide Intermediates, In Vitro Anticancer Screening, Structural Characterization, and Molecular Docking Study. | Branowska, D., et al. 2020. Molecules. 25: PMID: 32429377
- Synthesis, crystal structure and docking studies of tetracyclic 10-iodo-1,2-dihydroisoquinolino[2,1-b][1,2,4]benzothiadiazine 12,12-dioxide and its precursors. | Kolade, SO., et al. 2020. Acta Crystallogr C Struct Chem. 76: 810-820. PMID: 32756044
- Synthesis and Biological Evaluation of Novel Benzhydrylpiperazine-Coupled Nitrobenzenesulfonamide Hybrids. | Murthy, VS., et al. 2021. ACS Omega. 6: 9731-9740. PMID: 33869953
- Bacterial mutagenicity test data: collection by the task force of the Japan pharmaceutical manufacturers association. | Hakura, A., et al. 2021. Genes Environ. 43: 41. PMID: 34593056
- In vitro and in silico determination of glutaminyl cyclase inhibitors. | Tran, PT., et al. 2019. RSC Adv. 9: 29619-29627. PMID: 35531555
- First Fluorescent Acetylspermidine Deacetylation Assay for HDAC10 Identifies Selective Inhibitors with Cellular Target Engagement. | Herp, D., et al. 2022. Chembiochem. 23: e202200180. PMID: 35608330
- C-Sulfonylation of 4-Alkylpyridines: Formal Picolyl C-H Activation via Alkylidene Dihydropyridine Intermediates. | Tun, SL., et al. 2023. J Org Chem. 88: 3998-4002. PMID: 36848377
- The Synthesis and Transfection Activity of Disulfide Polycationic Amphiphiles. | Petukhov, IA., et al. 2023. Russ J Bioorg Chem. 49: 41-51. PMID: 37192981
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Nitrobenzenesulfonyl chloride, 25 g | sc-230595 | 25 g | $36.00 |
