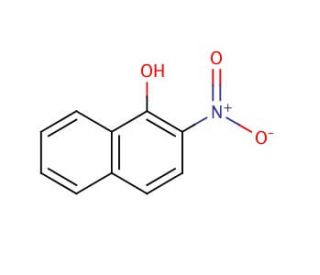
2-Nitro-1-naphthol (CAS 607-24-9)
QUICK LINKS
2-Nitro-1-naphthol is an organic compound attracting significant attention in the field of analytical chemistry. It can be converted to an amine for use in the synthesis of dyes, pigments, and other aromatic compounds with industrial applications. The naphthol moiety of 2-Nitro-1-naphthol is also a key functional group that can act as a chromophore; hence, it is frequently investigated in the development of colorimetric sensors that can detect metal ions or organic substances based on color change. Its ability to form complexes with metals is further utilized in the preparation of coordination compounds, which are of interest for their magnetic and electronic properties. 2-Nitro-1-naphthol is utilized as a starting material in the creation of heterocyclic compounds, providing a pathway to explore novel chemical space in the pursuit of new materials and chemical processes.
2-Nitro-1-naphthol (CAS 607-24-9) References
- The biochemistry of aromatic amines. 8. Synthesis and detection of di-(2-amino-1-naphthyl) hydrogen phosphate, a metabolite of 2-naphthylamine in dogs. | Boyland, E., et al. 1961. Biochem J. 78: 175-9. PMID: 16748865
- Human Toll-like receptor 8-selective agonistic activities in 1-alkyl-1H-benzimidazol-2-amines. | Beesu, M., et al. 2014. J Med Chem. 57: 7325-41. PMID: 25102141
- Light Absorption of Secondary Organic Aerosol: Composition and Contribution of Nitroaromatic Compounds. | Xie, M., et al. 2017. Environ Sci Technol. 51: 11607-11616. PMID: 28930472
- Characterization of aerosol nitroaromatic compounds: Validation of an experimental method. | Jaoui, M., et al. 2018. J Mass Spectrom. 53: 680-692. PMID: 29766603
- Stability and Efficiency of Mixed Aryl Phosphonate Prodrugs. | Foust, BJ., et al. 2019. ChemMedChem. 14: 1597-1603. PMID: 31226236
- Composition and light absorption of N-containing aromatic compounds in organic aerosols from laboratory biomass burning. | Xie, M., et al. 2019. Atmos Chem Phys. 19: 2899-2915. PMID: 31501655
- Roles of oxygen in photochemical reaction of naphthols in aqueous nitrite solution and mutagen formation. | Suzuki, J., et al. 1988. Chem Pharm Bull (Tokyo). 36: 4567-75. PMID: 3246026
- Composites and Copolymers Containing Redox-Active Molecules and Intrinsically Conducting Polymers as Active Masses for Supercapacitor Electrodes-An Introduction. | Holze, R. 2020. Polymers (Basel). 12: PMID: 32824366
- Secondary Formation of Aromatic Nitroderivatives of Environmental Concern: Photonitration Processes Triggered by the Photolysis of Nitrate and Nitrite Ions in Aqueous Solution. | Marussi, G. and Vione, D. 2021. Molecules. 26: PMID: 33925664
- Organic Small-Molecule Electrodes: Emerging Organic Composite Materials in Supercapacitors for Efficient Energy Storage. | He, Y., et al. 2022. Molecules. 27: PMID: 36431793
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Nitro-1-naphthol, 5 g | sc-230577 | 5 g | $146.00 |
