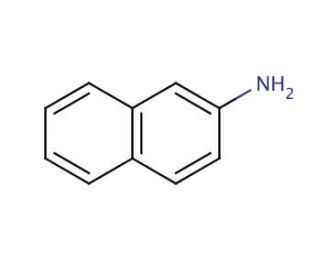
Molecular structure of 2-Naphthylamine, CAS Number: 91-59-8
2-Naphthylamine (CAS 91-59-8)
See product citations (1)
Alternate Names:
β-Naphthylamine
Application:
2-Naphthylamine is an amine compound used for research purposes
CAS Number:
91-59-8
Purity:
≥98%
Molecular Weight:
143.19
Molecular Formula:
C10H9N
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2-Naphthylamine is an aromatic amine used to make azo dyes. A common reason that it is used less for this purpose is that the substance is a carcinogen. Studies indicate that 2-Naphthylamine decreases the enzymatic functions of monoamine oxidase A and B. It is known that the main source of this compound is obtained from cigarette smoke. In addition, metabolites of 2-Naphthylamine such as N-hydroxy-2-Naphthylamine damages DNA by forming specific carcinogen-base adducts. These studies suggest that specific 2-Naphthylamine can cause problems for cells by binding to glycogen.
2-Naphthylamine (CAS 91-59-8) References
- 2-Naphthylamine, a compound found in cigarette smoke, decreases both monoamine oxidase A and B catalytic activity. | Hauptmann, N. and Shih, JC. 2001. Life Sci. 68: 1231-41. PMID: 11233991
- Determination of 2-naphthylamine in urine by a novel reversed-phase high-performance liquid chromatography method. | Hansen, AM., et al. 1992. J Chromatogr. 578: 85-90. PMID: 1400790
- Carcinogenicity of 2-naphthylhydroxylamine and 2-naphthylamine. | BOYLAND, E., et al. 1963. Br J Cancer. 17: 79-84. PMID: 14014726
- The carcinogenicity and metabolism of 2-naphthylamine and related compounds. | DEICHMANN, WB. and RADOMSKI, JL. 1963. Ind Med Surg. 32: 161-6. PMID: 14026492
- Spectroscopic investigation of the interaction of the toxicant, 2-naphthylamine, with bovine serum albumin. | Liu, Y., et al. 2011. J Biochem Mol Toxicol. 25: 362-8. PMID: 21800401
- Metabolic dephenylation of the rubber antioxidant N-phenyl-2-naphthylamine to carcinogenic 2-naphthylamine in rats. | Weiss, T., et al. 2013. Arch Toxicol. 87: 1265-72. PMID: 23423714
- 2-naphthylamine toxicity. | Czubacka, E. and Czerczak, S. 2020. Med Pr. 71: 205-220. PMID: 32118871
- [A new method for the determination of naphatylamines in workplace air for occupational exposure assessment]. | Jeżewska, A., et al. 2021. Med Pr. 72: 145-154. PMID: 33783437
- Alteration of urinary levels of the carcinogen, N-hydroxy-2-naphthylamine, and its N-glucuronide in the rat by control of urinary pH, inhibition of metabolic sulfation, and changes in biliary excretion. | Kadlubar, FF., et al. 1981. Chem Biol Interact. 33: 129-47. PMID: 6257402
- Local carcinogenicity, rates of absorption, extent and persistence of macromolecular binding, and acute histopathological effects of N-hydroxy-1-naphthylamine and N-hydroxy-2-naphthylamine. | Dooley, KL., et al. 1984. Cancer Res. 44: 1172-7. PMID: 6692401
- Activating and inactivating reactions controlling 2-naphthylamine mutagenicity. | Bock-Hennig, BS., et al. 1982. Arch Toxicol. 50: 259-66. PMID: 7149991
- Formation of urothelial and hepatic DNA adducts from carcinogen 2-naphthylamine. | Kadlubar, FF., et al. 1981. Carcinogenesis. 2: 467-70. PMID: 7273326
- Effect of pH on the neoplastic transformation of normal human skin fibroblasts by N-hydroxy-1-naphthylamine and N-hydroxy-2-naphthylamine. | Oldham, JW., et al. 1981. Carcinogenesis. 2: 937-40. PMID: 7296774
Inhibitor of:
TGase5.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Naphthylamine, 5 g | sc-209239 | 5 g | $218.00 |
