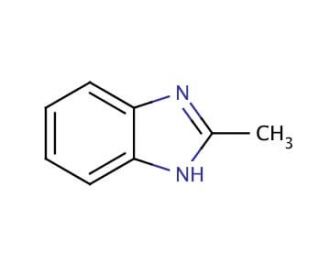
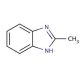
Molecular structure of 2-Methylbenzimidazole, CAS Number: 615-15-6
2-Methylbenzimidazole (CAS 615-15-6)
CAS Number:
615-15-6
Molecular Weight:
132.16
Molecular Formula:
C8H8N2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2-Methylbenzimidazole is a heterocyclic compound that is found in various biochemical processes. It is involved in the synthesis of various biomolecules and is an important building block in the formation of certain proteins and enzymes. 2-Methylbenzimidazole plays a role in the regulation of gene expression and is essential for the proper functioning of cells. 2-Methylbenzimidazole has antioxidant properties, which can help protect cells from damage caused by free radicals.
2-Methylbenzimidazole (CAS 615-15-6) References
- Heteroaromatic annulation of 2-methyl/2-cyanomethylbenzimidazole dianions with alpha-oxoketene dithioacetals: a highly regioselective synthetic protocol for 1,2- and 2,3-substituted/annulated pyrido[1,2-a]benzimidazoles. | Panda, K., et al. 2003. J Org Chem. 68: 3498-506. PMID: 12713352
- Syntheses and structures of two chiral zincophosphite compounds: [Zn(C8H8N2)(HPO3)] and (C6H13N2)[Zn3(C6H12N2)(HPO3)3(H2PO3)]. | Liu, L., et al. 2008. Dalton Trans. 2009-14. PMID: 18382778
- Cytotoxic activity, X-ray crystal structures and spectroscopic characterization of cobalt(II), copper(II) and zinc(II) coordination compounds with 2-substituted benzimidazoles. | Sánchez-Guadarrama, O., et al. 2009. J Inorg Biochem. 103: 1204-13. PMID: 19628280
- Molecular structure and vibrational spectra of 2- and 5-methylbenzimidazole molecules by density functional theory. | Güllüoglu, MT., et al. 2010. Spectrochim Acta A Mol Biomol Spectrosc. 76: 107-14. PMID: 20395166
- Above-room-temperature ferroelectricity and antiferroelectricity in benzimidazoles. | Horiuchi, S., et al. 2012. Nat Commun. 3: 1308. PMID: 23250438
- Quantum chemical assessment of benzimidazole derivatives as corrosion inhibitors. | Obayes, HR., et al. 2014. Chem Cent J. 8: 21. PMID: 24674343
- Stereoselective Alkylation of the Vinylketene Silyl N,O-Acetal and Its Application to the Synthesis of Mycocerosic Acid. | Nakamura, T., et al. 2016. Org Lett. 18: 132-5. PMID: 26673532
- Biofilm formation on copper and its control by inhibitor/biocide in cooling water environment. | Narenkumar, J., et al. 2021. Saudi J Biol Sci. 28: 7588-7594. PMID: 34867063
- Multivariate sodalite zeolitic imidazolate frameworks: a direct solvent-free synthesis. | López-Cabrelles, J., et al. 2022. Chem Sci. 13: 842-847. PMID: 35173949
- A convenient synthesis of N-(hetero)arylamides by the oxidative coupling of methylheteroarenes with amines. | Jaiswal, A., et al. 2022. Org Biomol Chem. 20: 6915-6922. PMID: 35979753
- Crystal Structure, Raman, FTIR, UV-Vis Absorption, Photoluminescence Spectroscopy, TG-DSC and Dielectric Properties of New Semiorganic Crystals of 2-Methylbenzimidazolium Perchlorate. | Balashova, E., et al. 2023. Materials (Basel). 16: PMID: 36903111
- Microbial transformation of nitroaromatic compounds in sewage effluent. | Hallas, LE. and Alexander, M. 1983. Appl Environ Microbiol. 45: 1234-41. PMID: 6859845
- Induction of chromosome loss in Saccharomyces cerevisiae strain D61.M by selected benzimidazole compounds. | Goin, CJ. and Mayer, VW. 1995. Mutat Res. 343: 185-99. PMID: 7623873
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Methylbenzimidazole, 5 g | sc-238144 | 5 g | $24.00 |
