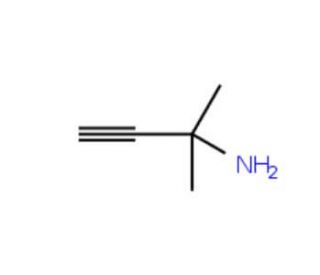
2-Methyl-3-butyn-2-amine (CAS 2978-58-7)
QUICK LINKS
2-Methyl-3-butyn-2-amine is an aliphatic amine compound that exhibits valuable properties for scientific research. Its role as a catalyst has been instrumental in polymer synthesis, while its reactivity as a reagent has enabled the synthesis of heterocyclic compounds. Moreover, 2-Methyl-3-butyn-2-amine has contributed to advancements in the synthesis of natural products and has played a significant role in the study of biological systems. The precise mechanism of action of 2-Methyl-3-butyn-2-amine is not yet entirely elucidated. However, it is believed to exhibit electrophilic reactivity, engaging with nucleophilic substrates to form new covalent bonds. Furthermore, its ability to form hydrogen bonds with other molecules is thought to play a role in various interactions.
2-Methyl-3-butyn-2-amine (CAS 2978-58-7) References
- Quaternary ammonium N,N-dichloroamines as topical, antimicrobial agents. | Francavilla, C., et al. 2009. Bioorg Med Chem Lett. 19: 2731-4. PMID: 19362467
- On terminal alkynes that can react with active-site cysteine nucleophiles in proteases. | Ekkebus, R., et al. 2013. J Am Chem Soc. 135: 2867-70. PMID: 23387960
- Straightforward strategy to substitute amide bonds by 1,2,3-triazoles in peptaibols analogs using Aibψ[Tz]-Xaa dipeptides. | Ben Haj Salah, K., et al. 2015. Biopolymers. 104: 611-21. PMID: 25784277
- Orally active ghrelin receptor inverse agonists and their actions on a rat obesity model. | Takahashi, B., et al. 2015. Bioorg Med Chem. 23: 4792-4803. PMID: 26100441
- Conformational promiscuity in triazolamers derived from quaternary amino acids mimics peptide behaviour. | Solà, J., et al. 2015. Org Biomol Chem. 13: 10797-801. PMID: 26455861
- Caspase-3 Substrates for Noninvasive Pharmacodynamic Imaging of Apoptosis by PET/CT. | Engel, BJ., et al. 2018. Bioconjug Chem. 29: 3180-3195. PMID: 30168713
- Silica-Supported Sterically Hindered Amines for CO2 Capture. | Lee, JJ., et al. 2018. Langmuir. 34: 12279-12292. PMID: 30244578
- Tailored therapeutics based on 1,2,3-1H-triazoles: a mini review. | Prasher, P. and Sharma, M. 2019. Medchemcomm. 10: 1302-1328. PMID: 31534652
- Green Conversion of CO2 and Propargylamines Triggered by Triply Synergistic Catalytic Effects in Metal-Organic Frameworks. | Jiang, XL., et al. 2021. Angew Chem Int Ed Engl. 60: 20417-20423. PMID: 34189807
- Discovery of Investigational Drug CT1812, an Antagonist of the Sigma-2 Receptor Complex for Alzheimer's Disease. | Rishton, GM., et al. 2021. ACS Med Chem Lett. 12: 1389-1395. PMID: 34531947
- Selectively Regulating Lewis Acid-Base Sites in Metal-Organic Frameworks for Achieving Turn-On/Off of the Catalytic Activity in Different CO2 Reactions. | Tian, XR., et al. 2022. Angew Chem Int Ed Engl. 61: e202200123. PMID: 35199447
- High-resolution mass spectrometry exhalome profiling with a modified direct analysis in real time ion source. | Xu, L., et al. 2022. Rapid Commun Mass Spectrom. 36: e9406. PMID: 36169592
- Synthesis of and radioligand binding studies with a tritiated pinacidil analogue: receptor interactions of structurally different classes of potassium channel openers and blockers. | Manley, PW., et al. 1993. J Med Chem. 36: 2004-10. PMID: 8336339
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Methyl-3-butyn-2-amine, 5 g | sc-230512 | 5 g | $49.00 |
