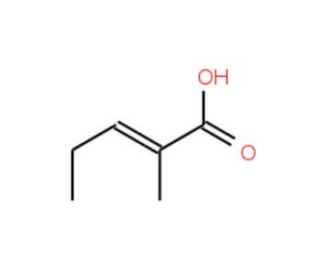
Molecular structure of 2-Methyl-2-pentenoic acid, CAS Number: 3142-72-1
2-Methyl-2-pentenoic acid (CAS 3142-72-1)
CAS Number:
3142-72-1
Molecular Weight:
114.15
Molecular Formula:
C6H10O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2-Methyl-2-pentenoic acid, a synthetic organic compound, is used in wastewater treatment. Classified as an alkanoic acid, it readily engages in acetylation to yield cinchonidine. 2-Methyl-2-pentenoic acid can be synthesized from pentenoic acid. it also has anti-inflammatory attributes, due to its ability to suppress fatty acid production via lipoxygenase enzyme inhibition. This compound also has immobilization properties and can be deuterated as a stable isotopic label.
2-Methyl-2-pentenoic acid (CAS 3142-72-1) References
- Identification of undescribed medium-chain acylcarnitines present in urine of patients with propionic and methylmalonic acidemias. | Libert, R., et al. 2000. Clin Chim Acta. 295: 87-96. PMID: 10767396
- Remote asymmetric induction with vinylketene silyl n,o-acetal. | Shirokawa, S., et al. 2004. J Am Chem Soc. 126: 13604-5. PMID: 15493903
- Effects of valproic acid derivatives on inositol trisphosphate depletion, teratogenicity, glycogen synthase kinase-3beta inhibition, and viral replication: a screening approach for new bipolar disorder drugs derived from the valproic acid core structure. | Eickholt, BJ., et al. 2005. Mol Pharmacol. 67: 1426-33. PMID: 15687223
- Hepatoprotection and lethality rescue by histone deacetylase inhibitor valproic acid in fatal hemorrhagic shock. | Gonzales, ER., et al. 2008. J Trauma. 65: 554-65. PMID: 18784568
- Safety and efficacy of 26 compounds belonging to chemical group 3 (α,β-unsaturated straight-chain and branched-chain aliphatic primary alcohols, aldehydes, acids and esters) when used as flavourings for all animal species and categories. | ,., et al. 2019. EFSA J. 17: e05654. PMID: 32626273
- Safety of 27 flavouring compounds providing a milky-vanilla flavour and belonging to different chemical groups for use as feed additives in all animal species (FEFANA asbl). | ,., et al. 2023. EFSA J. 21: e07713. PMID: 36698489
- The differences between broad bean koji fermented in laboratory and factory conditions by an efficient Aspergillus oryzae. | Li, H., et al. 2023. Front Microbiol. 14: 1139406. PMID: 37032872
- Reduction of 1, 1-dideuterio-2-methyl-2-penten-1-ol by beauveria sulfurescens. Mechanism of the microbiological reduction of α, β-unsaturated ald | Bostmembrun-Desrut, M., Dauphin, G., Kergomard, A., Renard, M. F., & Veschambre, H. 1985. Tetrahedron. 41(18): 3679-3682.
- Enantioselective hydrogenation of 2-methyl-2-pentenoic acid over cinchonidine-modified Pd/alumina | Borszeky, K., Mallat, T., & Baiker, A. 1996. Catalysis letters. 41: 199-202.
- Solvent and support effects in enantioselective hydrogenation of isophorone with (s)-α, α-diphenyl-2-pyrrolidinemethanol modified palladium | Sípos, É., Tungler, A., & Bitter, I. 2003. Reaction Kinetics and Catalysis Letters. 79: 101-109.
- Catecholamines and IL-4 synergize to induce arginase in myeloid suppressor cells after trauma | Popovic, P. J., Matta, B., Follansbee, C., & Ochoa, J. 2006. Journal of the American College of Surgeons. 203(3): S28-S29.
- An in situ spectroscopic study of prochiral reactant–chiral modifier interactions on palladium catalyst: case of alkenoic acid and cinchonidine in various solvents. | Tan, S., & Williams, C. T. 2013. The Journal of Physical Chemistry C. 117(35): 18043-18052.
- Elucidating Mammalian Anabolic Three‐Carbon Metabolism by Liquid Chromatography‐High Resolution Mass Spectrometry | Doan, M. T., Jiang, H., & Snyder, N. W. 2019. The FASEB Journal. 33(S1): 639.4-639.4.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Methyl-2-pentenoic acid, 10 g | sc-265785 | 10 g | $53.00 |
