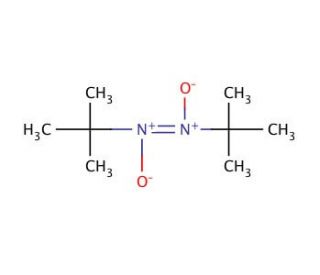
2-Methyl-2-nitrosopropane dimer (CAS 6841-96-9)
QUICK LINKS
2-Methyl-2-nitrosopropane dimer is often utilized in research focused on the study of nitroso compounds and their chemical behavior. This chemical is in the exploration of radical chemistry, particularly in understanding the formation and stability of nitroso dimer structures. In organic chemistry, 2-Methyl-2-nitrosopropane dimer is used to investigate mechanisms of nitrosation reactions, which are significant for synthesizing complex organic molecules. Additionally, the compound aids in the study of photochemical reactions where its properties can influence the understanding of light-induced processes in organic compounds. Moreover, its application in polymer research involves examining the effects of nitroso compounds on polymer degradation and stabilization, providing insights into improving material longevity and performance.
2-Methyl-2-nitrosopropane dimer (CAS 6841-96-9) References
- The role of the liver in the production of free radicals during halothane anaesthesia in the rat. Quantification of N-tert-butyl-alpha-(4- nitrophenyl)nitrone (PBN)-trapped adducts in bile from halothane as compared with carbon tetrachloride. | Hughes, HM., et al. 1991. Biochem J. 277 (Pt 3): 795-800. PMID: 1651704
- Chemical trapping of a primary quantum conversion product in photosynthesis. | Corker, GA., et al. 1966. Proc Natl Acad Sci U S A. 56: 1365-9. PMID: 16591382
- Exchange coupling mediated through-bonds and through-space in conformationally constrained polyradical scaffolds: calix[4]arene nitroxide tetraradicals and diradical. | Rajca, A., et al. 2006. J Am Chem Soc. 128: 13497-507. PMID: 17031963
- Free radical production from the interaction of 2-chloroethyl vesicants (mustard gas) with pyridine nucleotide-driven flavoprotein electron transport systems. | Brimfield, AA., et al. 2009. Toxicol Appl Pharmacol. 234: 128-34. PMID: 18977373
- Dye-incorporated coordination polymers for direct photocatalytic trifluoromethylation of aromatics at metabolically susceptible positions. | Zhang, T., et al. 2018. Nat Commun. 9: 4024. PMID: 30279417
- Direct introduction of nitrogen and oxygen functionality with spatial control using copper catalysis. | Shaum, JB., et al. 2018. Chem Sci. 9: 8748-8752. PMID: 30627395
- Identity of Low-Molecular-Weight Species Formed in End-To-End Cyclization Reactions Performed in THF. | Pan, CW., et al. 2018. Polymers (Basel). 10: PMID: 30960769
- Synthesis of Highly Congested Tertiary Alcohols via the [3,3] Radical Deconstruction of Breslow Intermediates. | Machín Rivera, R., et al. 2022. Org Lett. 24: 4275-4280. PMID: 35657720
- Research Progress in Energy Based on Polyphosphazene Materials in the Past Ten Years. | Zhou, Z., et al. 2022. Polymers (Basel). 15: PMID: 36616364
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Methyl-2-nitrosopropane dimer, 1 g | sc-251778 | 1 g | $74.00 |
