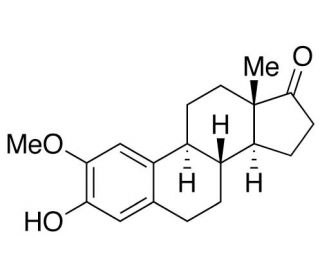
Molecular structure of 2-Methoxy Estrone, CAS Number: 362-08-3
2-Methoxy Estrone (CAS 362-08-3)
Alternate Names:
2-Hydroxyestrone 2-methyl ether
CAS Number:
362-08-3
Molecular Weight:
300.39
Molecular Formula:
C19H24O3
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2-Methoxy Estrone is a naturally occurring metabolite of the hormone estrogen. It is derived from estrone, which is one of the three major forms of estrogen produced in the human body. 2-Methoxyestrone is characterized by the addition of a methoxy group (-OCH3) at the 2-position of the estrone molecule.
2-Methoxy Estrone (CAS 362-08-3) References
- The conversion of 2-hydroxyestradiol-17 beta to 2-hydroxy and 2-methoxy metabolites in human urine. | AXELROD, LR., et al. 1961. Arch Biochem Biophys. 94: 265-8. PMID: 13685342
- 2-methoxyestrone as an estrogen metabolite in the human subject. | HOBKIRK, R. and NILSEN, M. 1963. J Clin Endocrinol Metab. 23: 274-8. PMID: 13954881
- Degradation of estradiol and ethinyl estradiol by activated sludge and by a defined mixed culture. | Weber, S., et al. 2005. Appl Microbiol Biotechnol. 67: 106-12. PMID: 15290133
- Estrogen metabolites in the release of inflammatory mediators from human amnion-derived cells. | Pavan, B., et al. 2011. Life Sci. 88: 551-8. PMID: 21277863
- Total dietary fat and omega-3 fatty acids have modest effects on urinary sex hormones in postmenopausal women. | Young, LR., et al. 2013. Nutr Metab (Lond). 10: 36. PMID: 23618064
- Qualitative and quantitative differences in estrogen biotransformation in human breast glandular and adipose tissues: implications for studies using mammary biospecimens. | Pemp, D., et al. 2019. Arch Toxicol. 93: 2823-2833. PMID: 31489452
- The metabolism of estradiol; oral compared to intravenous administration. | Longcope, C., et al. 1985. J Steroid Biochem. 23: 1065-70. PMID: 4094413
- Comparative fate of estrone and estrone sulfate in man. | Fishman, J. and Hellman, L. 1973. J Clin Endocrinol Metab. 36: 160-4. PMID: 4681879
- Estradiol transformation in men with breast cancer. | Zumoff, B., et al. 1966. J Clin Endocrinol Metab. 26: 960-6. PMID: 5926948
- Metabolism of radioactive estrone and estradiol by the golden hamster. | Collins, DC., et al. 1967. Endocrinology. 80: 893-5. PMID: 6023557
- Some ring a methoxy-derivatives of estrone and estradiol-17-beta. | Conrow, RB., et al. 1967. Steroids. 9: 307-26. PMID: 6034607
- Plasma precursors of estrogen. I. Extent of conversion of plasma delta-4-androstenedione to estrone in normal males and nonpregnant normal, castrate and adrenalectomized females. | MacDonald, PC., et al. 1967. J Clin Endocrinol Metab. 27: 1103-11. PMID: 6035658
- Metabolism of estrone sulphate by benign prostatic hyperplasia in vitro. | Carlström, K., et al. 1980. Scand J Urol Nephrol. 14: 135-7. PMID: 6163200
- Separation of steroidal estrogens and their major unconjugated metabolites by high performance liquid chromatography. | Aten, RF., et al. 1982. J Steroid Biochem. 16: 447-9. PMID: 6283270
- Estrogen 2-, 4-, 6- or 16-hydroxylation by human follicles shown by gas chromatography-mass spectrometry associated with stable isotope dilution. | Dehennin, L., et al. 1984. J Steroid Biochem. 20: 465-71. PMID: 6708529
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Methoxy Estrone, 50 mg | sc-482856 | 50 mg | $416.00 |
