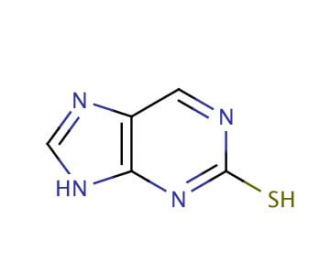
Molecular structure of 2-Mercaptopurine, CAS Number: 28128-19-0
2-Mercaptopurine (CAS 28128-19-0)
Alternate Names:
2-MP; 2-Purinethiol; 2-Thiopurine
Application:
2-Mercaptopurine is a thiopurine and analog of the anti-cancer drug 6-mercaptopurine (6-MP)
CAS Number:
28128-19-0
Molecular Weight:
152.18
Molecular Formula:
C5H4N4S
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2-Mercaptopurine is a purine analog as an antimetabolite. It is a potent inhibitor of the enzyme hypoxanthine-guanine phosphoribosyltransferase (HGPRT), which plays a role in the salvage pathway of purine nucleotide biosynthesis. By inhibiting this enzyme, 2-mercaptopurine disrupts the synthesis of DNA and RNA, ultimately leading to cell death. 2-Mercaptopurine is particularly useful in the study of purine metabolism and the development of anticancer.
2-Mercaptopurine (CAS 28128-19-0) References
- Kinetics of oxidation of aliphatic and aromatic thiols by myeloperoxidase compounds I and II. | Burner, U., et al. 1999. FEBS Lett. 443: 290-6. PMID: 10025950
- Tautomerism and infrared spectra of 2-thiopurine: an experimental matrix isolation and theoretical ab initio and density functional theory study. | Stepanenko, T., et al. 2001. Spectrochim Acta A Mol Biomol Spectrosc. 57: 375-83. PMID: 11206573
- Inhibition of 6-phosphofructo-2-kinase activity by mercaptopurines. | Mojena, M., et al. 1992. Biochem Pharmacol. 43: 671-8. PMID: 1311587
- Development of a markerless genetic exchange method for Methanosarcina acetivorans C2A and its use in construction of new genetic tools for methanogenic archaea. | Pritchett, MA., et al. 2004. Appl Environ Microbiol. 70: 1425-33. PMID: 15006762
- Development of a method for markerless genetic exchange in Enterococcus faecalis and its use in construction of a srtA mutant. | Kristich, CJ., et al. 2005. Appl Environ Microbiol. 71: 5837-49. PMID: 16204495
- Nature and position of functional group on thiopurine substrates influence activity of xanthine oxidase--enzymatic reaction pathways of 6-mercaptopurine and 2-mercaptopurine are different. | Tamta, H., et al. 2007. Biochemistry (Mosc). 72: 170-7. PMID: 17367294
- Overexpression of P-glycoprotein induces acquired resistance to imatinib in chronic myelogenous leukemia cells. | Peng, XX., et al. 2012. Chin J Cancer. 31: 110-8. PMID: 22098951
- Up-regulation of P-glycoprotein confers acquired resistance to 6-mercaptopurine in human chronic myeloid leukemia cells. | Peng, XX., et al. 2011. Oncol Lett. 2: 549-556. PMID: 22866119
- Functional identification of the hypoxanthine/guanine transporters YjcD and YgfQ and the adenine transporters PurP and YicO of Escherichia coli K-12. | Papakostas, K., et al. 2013. J Biol Chem. 288: 36827-40. PMID: 24214977
- Support of acute lymphoblastic leukemia cells by nonmalignant bone marrow stromal cells. | Usmani, S., et al. 2019. Oncol Lett. 17: 5039-5049. PMID: 31186715
- High-Throughput Fluorescent Assay for Inhibitor Screening of Proteases from RNA Viruses. | Cihlova, B., et al. 2021. Molecules. 26: PMID: 34206406
- In Silico Prediction of Metabolic Reaction Catalyzed by Human Aldehyde Oxidase. | Huang, M., et al. 2023. Metabolites. 13: PMID: 36984889
- Transient-state and steady-state kinetics of the oxidation of aliphatic and aromatic thiols by horseradish peroxidase. | Burner, U. and Obinger, C. 1997. FEBS Lett. 411: 269-74. PMID: 9271219
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Mercaptopurine, 25 mg | sc-206467 | 25 mg | $61.00 | |||
2-Mercaptopurine, 100 mg | sc-206467A | 100 mg | $191.00 |
