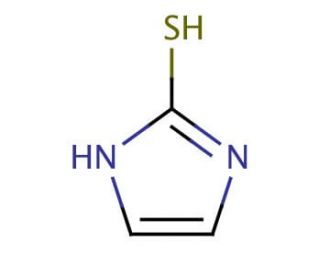
2-Mercaptoimidazole (CAS 872-35-5)
See product citations (1)
QUICK LINKS
2-Mercaptoimidazole is a compound that functions as a chelating agent in development applications. It acts by forming coordination complexes with metal ions, particularly copper and zinc, at the molecular level. This interaction allows 2-Mercaptoimidazole to effectively sequester metal ions, preventing their participation in various chemical reactions. 2-Mercaptoimidazole′s mechanism of action involves the formation of stable complexes with metal ions, thereby inhibiting their catalytic activity in experimental systems. By chelating metal ions, 2-Mercaptoimidazole may influence the outcome of chemical reactions and biochemical processes, which may be useful for studying metal ion-dependent pathways and mechanisms. Its ability to interact with metal ions at the molecular level makes 2-Mercaptoimidazole a component in experimental systems where the regulation of metal ion activity is of interest.
2-Mercaptoimidazole (CAS 872-35-5) References
- Design of anti-thyroid drugs: Binding studies and structure determination of the complex of lactoperoxidase with 2-mercaptoimidazole at 2.30 Å resolution. | Sirohi, HV., et al. 2017. Proteins. 85: 1882-1890. PMID: 28653416
- Post-Synthetic Mannich Chemistry on Metal-Organic Frameworks: System-Specific Reactivity and Functionality-Triggered Dissolution. | Amer Hamzah, H., et al. 2018. Chemistry. 24: 11094-11102. PMID: 29808942
- Repurposing the Pummerer Rearrangement: Determination of Methionine Sulfoxides in Peptides. | Woodroofe, CC., et al. 2020. Chembiochem. 21: 508-516. PMID: 31365170
- Strategy for Encapsulation of CdS Quantum Dots into Zeolitic Imidazole Frameworks for Photocatalytic Activity. | Son, YR., et al. 2020. Nanomaterials (Basel). 10: PMID: 33322795
- Methimazole in the Treatment of Melasma: A Clinical and Dermascopic Study. | Farag, A., et al. 2021. J Clin Aesthet Dermatol. 14: 14-20. PMID: 34221222
- The efficiency and safety of methimazole and propylthiouracil in hyperthyroidism: A meta-analysis of randomized controlled trials. | Tan, S., et al. 2021. Medicine (Baltimore). 100: e26707. PMID: 34397700
- Role of CYP2A6 in Methimazole Bioactivation and Hepatotoxicity. | Li, J., et al. 2021. Chem Res Toxicol. 34: 2534-2539. PMID: 34788025
- Review on Tailoring PEDOT:PSS Layer for Improved Device Stability of Perovskite Solar Cells. | Xia, Y., et al. 2021. Nanomaterials (Basel). 11: PMID: 34835883
- Synthesis of novel series of 3,5-disubstituted imidazo[1,2-d] [1,2,4]thiadiazoles involving SNAr and Suzuki-Miyaura cross-coupling reactions. | Pescheteau, C., et al. 2022. RSC Adv. 12: 6303-6313. PMID: 35424561
- Nanomaterials-Based Combinatorial Therapy as a Strategy to Combat Antibiotic Resistance. | León-Buitimea, A., et al. 2022. Antibiotics (Basel). 11: PMID: 35740200
- Safe and Effective Antioxidant: The Biological Mechanism and Potential Pathways of Ergothioneine in the Skin. | Liu, HM., et al. 2023. Molecules. 28: PMID: 36838636
- Comparison of the safety between propylthiouracil and methimazole with hyperthyroidism in pregnancy: A systematic review and meta-analysis. | Liu, Y., et al. 2023. PLoS One. 18: e0286097. PMID: 37205692
- Study of the Mechanism and Kinetics of the Radical Scavenging Activity of 2-Mercaptoimidazole. | Vo, QV., et al. 2023. J Phys Chem A. 127: 4934-4939. PMID: 37265437
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Mercaptoimidazole, 1 g | sc-238107 | 1 g | $41.00 |
