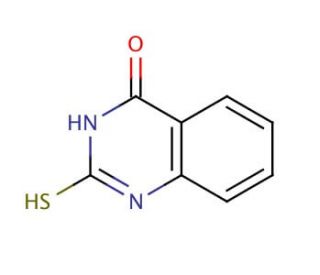
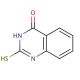
Molecular structure of 2-Mercapto-4(3H)-quinazolinone, CAS Number: 13906-09-7
2-Mercapto-4(3H)-quinazolinone (CAS 13906-09-7)
Alternate Names:
2-Thio-4(3H)-quinazolinone
Application:
2-Mercapto-4(3H)-quinazolinone is a quinazolinone derivative
CAS Number:
13906-09-7
Purity:
97%
Molecular Weight:
178.21
Molecular Formula:
C8H6N2OS
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2-Mercapto-4(3H)-quinazolinone is a thiol-containing quinazolinone derivative. 2-Mercapto-4(3H)-quinazolinone was described in a study of the human purine nucleoside phosphorylase enzyme, where it was used to generate a binary complex with the enzyme to produce a crystal for structural study. 2-Mercapto-4(3H)-quinazolinone was also demonstrated to produce inhibition of xanthine oxidase which was used to monitor free radical scavenging properties of this and other compounds.
2-Mercapto-4(3H)-quinazolinone (CAS 13906-09-7) References
- Reactive oxygen scavenger effect of pyrimidines, benzotriazoles and related compounds. | Lin, CY., et al. 2002. Anticancer Res. 22: 937-42. PMID: 12014675
- Structural studies of human purine nucleoside phosphorylase: towards a new specific empirical scoring function. | Timmers, LF., et al. 2008. Arch Biochem Biophys. 479: 28-38. PMID: 18790691
- The spectroscopic (FT-IR, FT-Raman and NMR), first order hyperpolarizability and HOMO-LUMO analysis of 2-mercapto-4(3H)-quinazolinone. | Prabavathi, N. and Senthil Nayaki, N. 2014. Spectrochim Acta A Mol Biomol Spectrosc. 129: 572-83. PMID: 24815813
- Design, synthesis of 2,3-disubstitued 4(3H)-quinazolinone derivatives as anti-inflammatory and analgesic agents: COX-1/2 inhibitory activities and molecular docking studies. | Abdel-Aziz, AA., et al. 2016. Bioorg Med Chem. 24: 3818-28. PMID: 27344214
- 2-Mercapto-Quinazolinones as Inhibitors of Type II NADH Dehydrogenase and Mycobacterium tuberculosis: Structure-Activity Relationships, Mechanism of Action and Absorption, Distribution, Metabolism, and Excretion Characterization. | Murugesan, D., et al. 2018. ACS Infect Dis. 4: 954-969. PMID: 29522317
- Synthesis and evaluation of 2-substituted 4(3H)-quinazolinone thioether derivatives as monoamine oxidase inhibitors. | Qhobosheane, MA., et al. 2018. Bioorg Med Chem. 26: 5531-5537. PMID: 30279044
- Insights into plasmon induced keto-enol isomerization. | Zhang, W., et al. 2020. Nanoscale. 12: 4334-4340. PMID: 32044913
- S-substituted 2-mercaptoquinazolin-4(3H)-one and 4-ethylbenzensulfonamides act as potent and selective human carbonic anhydrase IX and XII inhibitors. | El-Azab, AS., et al. 2020. J Enzyme Inhib Med Chem. 35: 733-743. PMID: 32189526
- Antibacterial activity evaluation of pleuromutilin derivatives with 4(3H)-quinazolinone scaffold against methicillin-resistant Staphylococcusaureus. | Deng, Y., et al. 2023. Eur J Med Chem. 246: 114960. PMID: 36462445
- Eco-friendly rapid synthesis of 3-substituted-2-thioxo-2,3-dihydroquinazolin-4(1H)-ones in choline chloride based deep eutectic solvent | Maja Molnar, Jelena Klenkar & Tena Tarnai. 2017. Synthetic Communications. 47: 1040-1045.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Mercapto-4(3H)-quinazolinone, 10 g | sc-225438 | 10 g | $124.00 |
