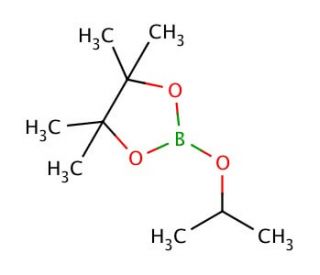
2-Isopropoxy-4,4,5,5-tetramethyl-1,3,2-dioxaborolane (CAS 61676-62-8)
QUICK LINKS
2-Isopropoxy-4,4,5,5-tetramethyl-1,3,2-dioxaborolane is a chemical reagent predominantly utilized in organic synthesis, particularly in the Suzuki coupling reaction, which is a palladium-catalyzed cross-coupling process that forms carbon-carbon bonds. This compound acts as a boron source, providing a stable but reactive partner for the cross-coupling with aryl or vinyl halides. Its stability under normal conditions and reactivity under catalytic conditions make it useful for chemists looking to build complex organic structures. Additionally, it is involved in the synthesis of biologically active molecules, serving as an intermediate for the introduction of boronic acid functionalities essential for further derivatizations.
2-Isopropoxy-4,4,5,5-tetramethyl-1,3,2-dioxaborolane (CAS 61676-62-8) References
- Precision synthesis of poly(3-hexylthiophene) from catalyst-transfer Suzuki-Miyaura coupling polymerization. | Yokozawa, T., et al. 2011. Macromol Rapid Commun. 32: 801-6. PMID: 21509845
- An Alternating Copolymer Derived from Indolo[3,2-b]carbazole and 4,7-Di(thieno[3,2-b]thien-2-yl)-2,1,3-benzothiadiazole for Photovoltaic Cells. | Xia, Y., et al. 2010. Macromol Rapid Commun. 31: 1287-92. PMID: 21567526
- Post-deposition treatment of an arylated-carbazole conjugated polymer for solar cell fabrication. | Liu, X., et al. 2012. Adv Mater. 24: 4505-10. PMID: 22718397
- Investigation of a lithium-halogen exchange flow process for the preparation of boronates by using a cryo-flow reactor. | Newby, JA., et al. 2014. Chemistry. 20: 263-71. PMID: 24339222
- Covalent Grafting of BPin functions on Carbon Nanotubes and Chan-Lam-Evans Post-Functionalization. | Desmecht, A., et al. 2019. Chemistry. 25: 1436-1440. PMID: 30325086
- A Novel Approach to Functionalization of Aryl Azides through the Generation and Reaction of Organolithium Species Bearing Masked Azides in Flow Microreactors. | Ichinari, D., et al. 2020. Angew Chem Int Ed Engl. 59: 1567-1571. PMID: 31733010
- Efficient preparation of 2-azulenylboronate and Miyaura-Suzuki cross-coupling reaction with aryl bromides for easy access to poly (2-azulenyl) benzenes | Ito, S., Terazono, T., Kubo, T., Okujima, T., Morita, N., Murafuji, T.,.. & Tajiri, A. 2004. Tetrahedron. 60(25): 5357-5366.
- Highly luminescent 1, 4-diketo-3, 6-diphenylpyrrolo [3, 4-c] pyrrole-(DPP-) based conjugated polymers prepared upon Suzuki coupling | Zhu, Y., Rabindranath, A. R., Beyerlein, T., & Tieke, B. 2007. Macromolecules. 40(19): 6981-6989.
- Mild and Selective Synthesis of an Aryl Boronic Ester by Equilibration of Mixtures of Boronic and Borinic Acid Derivatives | Hawkins, V. F., Wilkinson, M. C., & Whiting, M. 2008. Organic Process Research & Development. 12(6): 1265-1268.
- Preparative synthesis via continuous flow of 4, 4, 5, 5-tetramethyl-2-(3-trimethylsilyl-2-propynyl)-1, 3, 2-dioxaborolane: a general propargylation reagent. | Fandrick, D. R., Roschangar, F., Kim, C., Hahm, B. J., Cha, M. H., Kim, H. Y.,.. & Senanayake, C. H. 2012. Organic Process Research & Development. 16(5): 1131-1140.
- An innovative and efficient synthesis of stable isotope labelled 1‐methyl‐5‐(4, 4, 5, 5‐tetramethyl‐1, 3, 2‐dioxaborolan‐2‐yl)‐1H‐pyrazole via [13C42H3] N‐methylpyrazole | Manning, C., Athlan, A., & Badman, G. 2012. Journal of Labelled Compounds and Radiopharmaceuticals. 55(13): 467-469.
- A novel approach for the synthesis of Crizotinib through the key chiral alcohol intermediate by asymmetric hydrogenation using highly active Ir-Spiro-PAP catalyst | Qian, J. Q., Yan, P. C., Che, D. Q., Zhou, Q. L., & Li, Y. Q. 2014. Tetrahedron Letters. 55(9): 1528-1531.
- Synthesis of Biaryls Having a Piperidylmethyl Group Based on Space Integration of Lithiation, Borylation, and Suzuki–Miyaura Coupling | Takahashi, Y., Ashikari, Y., Takumi, M., Shimizu, Y., Jiang, Y., Higuma, R.,.. & Nagaki, A. 2020. European Journal of Organic Chemistry. 2020(5): 618-622.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Isopropoxy-4,4,5,5-tetramethyl-1,3,2-dioxaborolane, 5 ml | sc-256167 | 5 ml | $42.00 | |||
2-Isopropoxy-4,4,5,5-tetramethyl-1,3,2-dioxaborolane, 25 ml | sc-256167A | 25 ml | $77.00 |
