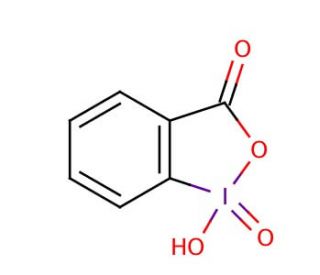
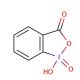
2-Iodoxybenzoic acid (CAS 61717-82-6)
QUICK LINKS
2-Iodoxybenzoic acid is a powerful oxidizing agent that is in organic synthesis as a reagent for the conversion of alcohols to aldehydes or ketones. It functions by transferring an oxygen atom to the alcohol, resulting in the formation of the carbonyl compound. 2-Iodoxybenzoic Acid′s mechanism of action involves the transfer of the oxygen atom from the iodoxy group to the alcohol, leading to the formation of the corresponding carbonyl compound and iodosobenzoic acid as a byproduct. The reaction proceeds through a series of electron transfer steps, ultimately resulting in the oxidation of the alcohol. 2-Iodoxybenzoic Acid′s ability to selectively oxidize alcohols to carbonyl compounds may be useful in the synthesis of various organic molecules. Its mechanism of action at the molecular level involves the transfer of oxygen atoms, enabling the conversion of alcohols to aldehydes or ketones in a controlled and efficient manner.
2-Iodoxybenzoic acid (CAS 61717-82-6) References
- A stabilized formulation of IBX (SIBX) for safe oxidation reactions including a new oxidative demethylation of phenolic methyl aryl ethers. | Ozanne, A., et al. 2003. Org Lett. 5: 2903-6. PMID: 12889904
- o-Iodoxybenzoic acid (IBX) as a viable reagent in the manipulation of nitrogen- and sulfur-containing substrates: scope, generality, and mechanism of IBX-mediated amine oxidations and dithiane deprotections. | Nicolaou, KC., et al. 2004. J Am Chem Soc. 126: 5192-201. PMID: 15099102
- Synthesis of the catechols of natural and synthetic estrogens by using 2-iodoxybenzoic acid (IBX) as the oxidizing agent. | Saeed, M., et al. 2005. Steroids. 70: 173-8. PMID: 15763595
- Esters of 2-iodoxybenzoic acid: hypervalent iodine oxidizing reagents with a pseudobenziodoxole structure. | Zhdankin, VV., et al. 2005. J Org Chem. 70: 6484-91. PMID: 16050713
- Enhancing 2-iodoxybenzoic acid reactivity by exploiting a hypervalent twist. | Su, JT. and Goddard, WA. 2005. J Am Chem Soc. 127: 14146-7. PMID: 16218584
- New synthetic approach for the construction of multisubstituted 2-acyl furans by the IBX-mediated cascade oxidation/cyclization of cis-2-En-4-yn-1-ols (IBX=2-iodoxybenzoic acid). | Du, X., et al. 2008. Chemistry. 14: 9495-8. PMID: 18810734
- A selective de-O-methylation of guaiacyl lignans to corresponding catechol derivatives by 2-iodoxybenzoic acid (IBX). The role of the catechol moiety on the toxicity of lignans. | Bernini, R., et al. 2009. Org Biomol Chem. 7: 2367-77. PMID: 19462048
- New lipophilic piceatannol derivatives exhibiting antioxidant activity prepared by aromatic hydroxylation with 2-iodoxybenzoic acid (IBX). | Bernini, R., et al. 2009. Molecules. 14: 4669-81. PMID: 19924094
- Room-temperature aromatization of tetrahydro-β-carbolines by 2-iodoxybenzoic acid: utility in a total synthesis of eudistomin U. | Panarese, JD. and Waters, SP. 2010. Org Lett. 12: 4086-9. PMID: 20715768
- 2-Iodoxybenzoic acid--a simple oxidant with a dazzling array of potential applications. | Duschek, A. and Kirsch, SF. 2011. Angew Chem Int Ed Engl. 50: 1524-52. PMID: 21271626
- 2-Iodoxybenzoic acid organosulfonates: preparation, X-ray structure and reactivity of new, powerful hypervalent iodine(V) oxidants. | Yusubov, MS., et al. 2013. Chem Commun (Camb). 49: 11269-71. PMID: 24153437
- Regioselective dehydrogenation of 3-keto-steroids to form conjugated enones using o-iodoxybenzoic acid and trifluoroacetic acid catalysis. | Iida, T., et al. 2014. Chem Phys Lipids. 178: 45-51. PMID: 24309193
- A Twist of the Twist Mechanism, 2-Iodoxybenzoic Acid (IBX)-Mediated Oxidation of Alcohol Revisited: Theory and Experiment. | Jiang, H., et al. 2017. Org Lett. 19: 6502-6505. PMID: 29166031
- 2-Iodoxybenzoic acid ditriflate: the most powerful hypervalent iodine(v) oxidant. | Yusubov, MS., et al. 2019. Chem Commun (Camb). 55: 7760-7763. PMID: 31209449
- Practical Synthesis of 2-Iodosobenzoic Acid (IBA) without Contamination by Hazardous 2-Iodoxybenzoic Acid (IBX) under Mild Conditions. | China, H., et al. 2021. Molecules. 26: PMID: 33801611
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Iodoxybenzoic acid, 1 g | sc-256165 | 1 g | $34.00 | |||
2-Iodoxybenzoic acid, 10 g | sc-256165A | 10 g | $173.00 |
