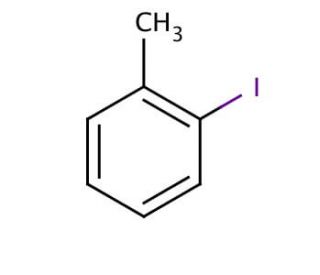
2-Iodotoluene (CAS 615-37-2)
QUICK LINKS
2-Iodotoluene is a chemical compound that functions as a substrate in various organic synthesis reactions. It acts as an electrophile in aromatic substitution reactions, where it undergoes substitution with nucleophiles to form new carbon-carbon or carbon-heteroatom bonds. 2-Iodotoluene′s mechanism of action involves participating in reactions such as Suzuki coupling, Heck reaction, and Buchwald-Hartwig amination, where it serves as a precursor for the introduction of the iodine atom into the target molecule. Through its involvement in these processes, 2-Iodotoluene contributes to the formation of complex organic molecules with specific structural and functional properties. Its role in these reactions allows for the creation of diverse chemical compounds with potential applications in materials science, chemical biology, and other areas.
2-Iodotoluene (CAS 615-37-2) References
- Structures, biological activities, and total syntheses of 13-hydroxy- and 13-acetoxy-14-nordehydrocacalohastine, novel modified furanoeremophilane-type sesquiterpenes from Trichilia cuneata. | Doe, M., et al. 2005. Org Lett. 7: 1765-8. PMID: 15844901
- Synthesis of benzannulated N-heterocycles by a palladium-catalyzed C-C/C-N coupling of bromoalkylamines. | Thansandote, P., et al. 2007. Org Lett. 9: 5255-8. PMID: 18001046
- Copper-catalyzed guanidinylation of aryl iodides: the formation of N,N'-disubstituted guanidines. | Cortes-Salva, M., et al. 2010. Org Lett. 12: 1316-9. PMID: 20170103
- Expeditious synthesis of phenanthridines from benzylamines via dual palladium catalysis. | Maestri, G., et al. 2010. Org Lett. 12: 5692-5. PMID: 21082784
- Ligand-free copper-catalyzed arylation of amidines. | Cortes-Salva, M., et al. 2011. J Org Chem. 76: 1456-9. PMID: 21250705
- Ruthenium-catalyzed carbonylative C-C coupling in water by directed C-H bond activation. | Tlili, A., et al. 2013. Angew Chem Int Ed Engl. 52: 6293-7. PMID: 23629872
- Green synthesis of Pd nanoparticles mediated by Euphorbia thymifolia L. leaf extract: Catalytic activity for cyanation of aryl iodides under ligand-free conditions. | Nasrollahzadeh, M. and Sajadi, SM. 2016. J Colloid Interface Sci. 469: 191-195. PMID: 26890384
- Comparative profiling of well-defined copper reagents and precursors for the trifluoromethylation of aryl iodides. | Kaplan, PT., et al. 2017. Beilstein J Org Chem. 13: 2297-2303. PMID: 29181108
- Palladium-Catalyzed trans-Hydroalkoxylation: Counterintuitive Use of an Aryl Iodide Additive to Promote C-H Bond Formation. | Das, A., et al. 2022. ACS Catal. 12: 7565-7570. PMID: 35799768
- Synthetic strategies for the preparation of γ-phostams: 1,2-azaphospholidine 2-oxides and 1,2-azaphospholine 2-oxides. | Xu, J. 2022. Beilstein J Org Chem. 18: 889-915. PMID: 35957753
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Iodotoluene, 5 g | sc-230437 | 5 g | $20.00 |
