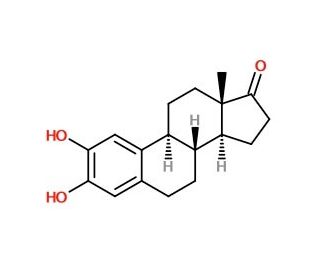
Molecular structure of 2-Hydroxy Estrone, CAS Number: 362-06-1
2-Hydroxy Estrone (CAS 362-06-1)
See product citations (1)
Alternate Names:
Catecholestrone
Application:
2-Hydroxy Estrone is an estradiol metabolite
CAS Number:
362-06-1
Molecular Weight:
286.37
Molecular Formula:
C18H22O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2-Hydroxy Estrone is a naturally occurring metabolite of estradiol hormone. It plays a role in regulating estrogen-dependent processes. Researchers have identified its significance as one of the primary metabolites of estradiol. Studies have explored the potential of 2-Hydroxy Estrone as a biomarker for various applications.Researchers have employed 2-Hydroxy Estrone to examine the effects of estrogen on the cardiovascular and immune systems.The mechanism of action of 2-Hydroxy Estroneinvolves its role as an estrogen receptor agonist. It binds to the receptor and activates it, thereby influencing a variety of estrogen-dependent processes.
2-Hydroxy Estrone (CAS 362-06-1) References
- Reproducibility over time of measurements of androgens, estrogens and hydroxy estrogens in urine samples from post-menopausal women. | Rinaldi, S., et al. 2003. Eur J Epidemiol. 18: 417-24. PMID: 12889688
- Estrogen metabolites in the release of inflammatory mediators from human amnion-derived cells. | Pavan, B., et al. 2011. Life Sci. 88: 551-8. PMID: 21277863
- Total dietary fat and omega-3 fatty acids have modest effects on urinary sex hormones in postmenopausal women. | Young, LR., et al. 2013. Nutr Metab (Lond). 10: 36. PMID: 23618064
- Covalent binding to proteins of reactive intermediates resulting from prostaglandin H synthase-catalyzed oxidation of stilbene and steroid estrogens. | Freyberger, A. and Degen, GH. 1989. J Biochem Toxicol. 4: 95-103. PMID: 2512390
- Novel and potent biological antioxidants on membrane phospholipid peroxidation: 2-hydroxy estrone and 2-hydroxy estradiol. | Nakano, M., et al. 1987. Biochem Biophys Res Commun. 142: 919-24. PMID: 3827906
- Estrogen 2-, 4-, 6- or 16-hydroxylation by human follicles shown by gas chromatography-mass spectrometry associated with stable isotope dilution. | Dehennin, L., et al. 1984. J Steroid Biochem. 20: 465-71. PMID: 6708529
- Role of catechol estrogens in activation of lordosis in female rats and guinea pigs. | Marrone, BL., et al. 1977. Pharmacol Biochem Behav. 7: 13-7. PMID: 905327
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Hydroxy Estrone, 1 mg | sc-206452 | 1 mg | $286.00 | |||
2-Hydroxy Estrone, 1 mg | sc-206452-CW | 1 mg | $439.00 | |||
2-Hydroxy Estrone, 2 mg | sc-206452A | 2 mg | $561.00 | |||
2-Hydroxy Estrone, 5 mg | sc-206452B | 5 mg | $1361.00 | |||
2-Hydroxy Estrone, 10 mg | sc-206452C | 10 mg | $2336.00 | |||
2-Hydroxy Estrone, 20 mg | sc-206452D | 20 mg | $4080.00 |
