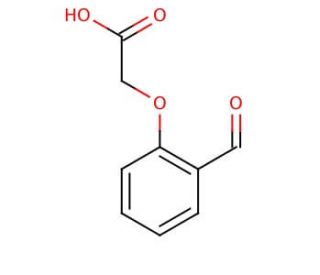
2-Formylphenoxyacetic acid (CAS 6280-80-4)
QUICK LINKS
2-Formylphenoxyacetic acid is an organic compound classified as a phenoxyacetic acid. It holds significant importance as an intermediate compound in the synthesis of various compounds. Several synthetic pathways exist for the production of 2-Formylphenoxyacetic acid, and its applications in scientific research are diverse. Additionally, 2-Formylphenoxyacetic acid finds utility in the creation of polymers such as polyphenols, as well as in the synthesis of polyamides, polyesters, dyes, and optical materials. As an intermediate compound, 2-Formylphenoxyacetic acid assumes a catalytic role in various reactions, leading to the formation of desired products. Although the precise mechanism of action remains incompletely understood, it is believed to participate in the establishment of a covalent bond between the reactants.
2-Formylphenoxyacetic acid (CAS 6280-80-4) References
- A self-replicating ligase ribozyme. | Paul, N. and Joyce, GF. 2002. Proc Natl Acad Sci U S A. 99: 12733-40. PMID: 12239349
- Synthesis, characterization and antibacterial activity of azomethine derivatives derived from 2-formylphenoxyacetic acid. | Iqbal, A., et al. 2007. Molecules. 12: 245-54. PMID: 17846575
- Synthesis and spectroscopic studies of new Schiff bases. | Siddiqui, HL., et al. 2006. Molecules. 11: 206-11. PMID: 17962791
- Design, synthesis and DNA binding activities of late first row transition metal(II) complexes of bi- functional tri - and tetratopic imines. | Netalkar, PP., et al. 2012. Spectrochim Acta A Mol Biomol Spectrosc. 97: 762-70. PMID: 22902573
- In situ Bronsted-Lowry acid catalyzed syntheses, characterization, single crystal XRD, electronic spectral-, DPPH radical scavenging-, and DNA protection studies of aryl-3,3'-bis(indolyl)methanes. | Suresh Kumar, GS., et al. 2014. Spectrochim Acta A Mol Biomol Spectrosc. 123: 249-56. PMID: 24398468
- Titania-promoted carboxylic acid alkylations of alkenes and cascade addition-cyclizations. | Manley, DW., et al. 2014. J Org Chem. 79: 1386-98. PMID: 24437519
- Molecular structure investigation of organic cocrystals of 1,10-phenanthroline-5,6-dione with aryloxyacetic acid: a combined experimental and theoretical study. | Suresh Kumar, GS., et al. 2014. Spectrochim Acta A Mol Biomol Spectrosc. 132: 465-76. PMID: 24887507
- Synthesis and antibacterial activity of Schiff bases and amines derived from alkyl 2-(2-formyl-4-nitrophenoxy)alkanoates. | Goszczyńska, A., et al. 2015. Med Chem Res. 24: 3561-3577. PMID: 26213456
- Metabolism of 4-chloro-2-methylphenoxyacetate by a soil pseudomonad. Preliminary evidence for the metabolic pathway. | Gaunt, JK. and Evans, WC. 1971. Biochem J. 122: 519-26. PMID: 5123885
- Effects of antibiotics on platelet functions in human plasma in vitro and dog plasma in vivo. | Genua, MI., et al. 1980. J Pharm Sci. 69: 1282-4. PMID: 7452456
- Binding of toxic metabolites of isoniazid by aconiazide. | Held, HR. and Landi, S. 1980. J Pharm Sci. 69: 1284-7. PMID: 7452457
- Pharmacokinetic evaluation of aconiazide, a potentially less toxic isoniazid prodrug. | Peloquin, CA., et al. 1994. Pharmacotherapy. 14: 415-23. PMID: 7937278
- Molecular cloning, expression and catalytic activity of a human AKR7 member of the aldo-keto reductase superfamily: evidence that the major 2-carboxybenzaldehyde reductase from human liver is a homologue of rat aflatoxin B1-aldehyde reductase. | Ireland, LS., et al. 1998. Biochem J. 332 (Pt 1): 21-34. PMID: 9576847
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Formylphenoxyacetic acid, 25 g | sc-230358 | 25 g | $79.00 |
