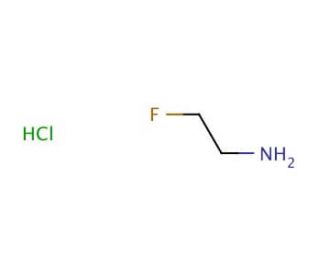
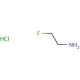
Molecular structure of 2-Fluoroethylamine hydrochloride, CAS Number: 460-08-2
2-Fluoroethylamine hydrochloride (CAS 460-08-2)
Alternate Names:
2-fluoroethan-1-amine hydrochloride
CAS Number:
460-08-2
Purity:
≥95%
Molecular Weight:
99.54
Molecular Formula:
C2H6FN•HCl
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2-Fluoroethylamine hydrochloride, or 2-Fluoroethanamine hydrochloride, is an amine compound that falls into the category of organic compounds. It is obtained through the reaction between 2-fluoroethanol and hydrogen chloride. This compound has been the subject of extensive scientific research due to its diverse biochemical effects and has shown promising applications in various fields.
2-Fluoroethylamine hydrochloride (CAS 460-08-2) References
- The observation of a large gauche preference when 2-fluoroethylamine and 2-fluoroethanol become protonated. | Briggs, CR., et al. 2004. Org Biomol Chem. 2: 732-40. PMID: 14985814
- Structure-based design of 7-carbamate analogs of geldanamycin. | Rastelli, G., et al. 2005. Bioorg Med Chem Lett. 15: 5016-21. PMID: 16165354
- Rapid exchange of fluoroethylamine via the Rhesus complex in human erythrocytes: 19F NMR magnetization transfer analysis showing competition by ammonia and ammonia analogues. | Szekely, D., et al. 2006. Biochemistry. 45: 9354-61. PMID: 16866382
- Conformational preferences and basicities of monofluorinated cyclopropyl amines in comparison to cyclopropylamine and 2-fluoroethylamine. | Hyla-Kryspin, I., et al. 2008. Org Biomol Chem. 6: 4167-75. PMID: 18972047
- Fluorine conformational effects in organocatalysis: an emerging strategy for molecular design. | Zimmer, LE., et al. 2011. Angew Chem Int Ed Engl. 50: 11860-71. PMID: 21953782
- Improving the capture of CO2 by substituted monoethanolamines: electronic effects of fluorine and methyl substituents. | Gangarapu, S., et al. 2012. Chemphyschem. 13: 3973-80. PMID: 22965750
- Conformational analysis of 2,2-difluoroethylamine hydrochloride: double gauche effect. | Silla, JM., et al. 2014. Beilstein J Org Chem. 10: 877-82. PMID: 24778743
- Direct Brønsted analysis of the restoration of activity to a mutant enzyme by exogenous amines. | Toney, MD. and Kirsch, JF. 1989. Science. 243: 1485-8. PMID: 2538921
- Amyloid beta protein enhances the survival of hippocampal neurons in vitro. | Whitson, JS., et al. 1989. Science. 243: 1488-90. PMID: 2928783
- Stereocontrolled Synthesis of Functionalized Azaheterocycles from Carbocycles through Oxidative Ring Opening/Reductive Ring Closing Protocols. | Kiss, L., et al. 2020. Chem Rec. 20: 120-141. PMID: 31250972
- Regulating Reversible Phase Transition Behaviors by Poly-H/F Substitution in Hybrid Perovskite-Like 2[CH2FCH2NH3]·[CdCl4]. | Song, N., et al. 2020. ACS Omega. 5: 6773-6780. PMID: 32258912
- Nature-inspired dimerization as a strategy to modulate neuropeptide pharmacology exemplified with vasopressin and oxytocin. | Dekan, Z., et al. 2021. Chem Sci. 12: 4057-4062. PMID: 34163676
- Strained Ammonium Precursors for Radiofluorinations. | Reissig, F. and Mamat, C. 2022. ChemistryOpen. 11: e202200039. PMID: 35736542
- Alkylating activity of 1,3-bis(2-chloroethyl)-1-nitrosourea and related compounds. | Wheeler, GP. and Chumley, S. 1967. J Med Chem. 10: 259-61. PMID: 6034073
- Studies on synthesis and anticancer activity of selected N-(2-fluoroethyl)-N-nitrosoureas. | Johnston, TP., et al. 1984. J Med Chem. 27: 1422-6. PMID: 6492072
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Fluoroethylamine hydrochloride, 1 g | sc-230346 | 1 g | $40.00 | |||
2-Fluoroethylamine hydrochloride, 5 g | sc-230346A | 5 g | $115.00 | |||
2-Fluoroethylamine hydrochloride, 10 g | sc-230346B | 10 g | $170.00 | |||
2-Fluoroethylamine hydrochloride, 25 g | sc-230346C | 25 g | $345.00 | |||
2-Fluoroethylamine hydrochloride, 100 g | sc-230346D | 100 g | $1252.00 |
