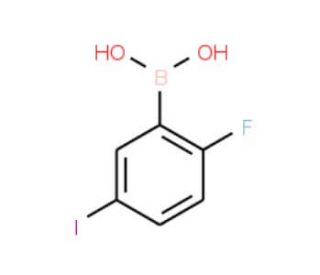
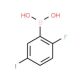
2-Fluoro-5-iodophenylboronic acid (CAS 866683-41-2)
QUICK LINKS
2-Fluoro-5-iodophenylboronic acid is a chemical compound that functions as a key building block in the synthesis of various organic molecules. Its mechanism of action involves participating in Suzuki-Miyaura cross-coupling reactions, where it acts as a boron source to form carbon-carbon bonds. 2-Fluoro-5-Iodophenylboronic Acid facilitates the formation of aryl and heteroaryl compounds, which are important in the development of agrochemicals, and materials science. Through its interaction at the molecular level, 2-Fluoro-5-iodophenylboronic acid enables the construction of complex molecular structures, allowing for the creation of diverse chemical entities with potential applications in various fields. Its role in organic synthesis contributes to the advancement of chemical science by providing a means to access novel compounds for further investigation and potential use in a range of experimental applications.
2-Fluoro-5-iodophenylboronic acid (CAS 866683-41-2) References
- Scope and Limitations of the Liebeskind-Srogl Cross-Coupling Reactions Involving the Biellmann BODIPY. | Betancourt-Mendiola, L., et al. 2015. J Org Chem. 80: 5771-82. PMID: 25965325
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Fluoro-5-iodophenylboronic acid, 1 g | sc-298449 | 1 g | $31.00 |
