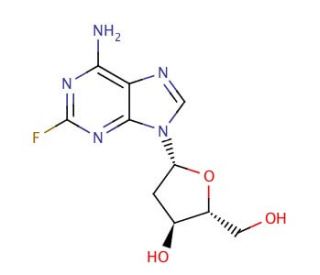
2-Fluoro-2′-deoxyadenosine (CAS 21679-12-9)
QUICK LINKS
2-Fluoro-2′-deoxyadenosine is a compound that is extensively utilized in biochemical research, particularly in the study of nucleic acid metabolism and function. It serves as an analog of deoxyadenosine, enabling researchers to investigate the impact of the fluorine substitution on enzymatic processes, such as those catalyzed by polymerases and nucleases. The compound is of particular interest in the exploration of DNA synthesis and repair mechanisms, as it can be incorporated into DNA strands in place of natural nucleotides to study the effects of altered base pairing stability and conformation. Additionally, 2-Fluoro-2′-deoxyadenosine is used in the field of molecular biology to probe the specificity of adenosine-related enzymes and to understand the dynamics of nucleotide flipping within DNA or RNA structures. Its incorporation into oligonucleotides also aids in the study of structural biology, providing insights into the influence of halogenated nucleotides on the three-dimensional architecture of nucleic acids.
2-Fluoro-2′-deoxyadenosine (CAS 21679-12-9) References
- Antitumor activity of 2-fluoro-2'-deoxyadenosine against tumors that express Escherichia coli purine nucleoside phosphorylase. | Parker, WB., et al. 2003. Cancer Gene Ther. 10: 23-9. PMID: 12489025
- A human biotin acceptor domain allows site-specific conjugation of an enzyme to an antibody-avidin fusion protein for targeted drug delivery. | Asai, T., et al. 2005. Biomol Eng. 21: 145-55. PMID: 15748688
- Study of a hammerhead ribozyme containing 2'-modified adenosine residues. | Olsen, DB., et al. 1991. Biochemistry. 30: 9735-41. PMID: 1911762
- Structure of a mutant human purine nucleoside phosphorylase with the prodrug, 2-fluoro-2'-deoxyadenosine and the cytotoxic drug, 2-fluoroadenine. | Afshar, S., et al. 2009. Protein Sci. 18: 1107-14. PMID: 19388075
- Atomic substitution reveals the structural basis for substrate adenine recognition and removal by adenine DNA glycosylase. | Lee, S. and Verdine, GL. 2009. Proc Natl Acad Sci U S A. 106: 18497-502. PMID: 19841264
- Susceptibility in vitro of clinically metronidazole-resistant Trichomonas vaginalis to nitazoxanide, toyocamycin, and 2-fluoro-2'-deoxyadenosine. | Wright, JM., et al. 2010. Parasitol Res. 107: 847-53. PMID: 20532912
- Structural and kinetic studies of Schistosoma mansoni adenylate kinases. | Marques, Ide A., et al. 2012. Mol Biochem Parasitol. 185: 157-60. PMID: 22841753
- Synthesis, solution and crystal structure of the coenzyme B(12) analogue Co(β)-2'-fluoro-2',5'-dideoxyadenosylcobalamin. | Hunger, M., et al. 2015. J Inorg Biochem. 148: 62-8. PMID: 25726330
- Characterization of 2-Fluoro-2'-deoxyadenosine in Duplex, G-Quadruplex and i-Motif. | Hirashima, S., et al. 2022. Chembiochem. 23: e202200222. PMID: 35438834
- Nitro-Activated Nucleobase Exchange in the Synthesis of 2'-Fluoro-2'-Deoxyribonucleosides. | Gong, Y., et al. 2022. J Org Chem. 87: 9330-9342. PMID: 35759615
- Isolation and Characterization of Engineered Nucleoside Deoxyribosyltransferase with Enhanced Activity Toward 2'-Fluoro-2'-Deoxynucleoside. | Yoo, YJ., et al. 2022. J Microbiol Biotechnol. 32: 1041-1046. PMID: 35791073
- Pathways of nucleotide metabolism in Schistosoma mansoni--VII. Inhibition of adenine and guanine nucleotide synthesis by purine analogs in intact worms. | Senft, AW. and Crabtree, GW. 1977. Biochem Pharmacol. 26: 1847-55. PMID: 410421
- Multinuclear magnetic resonance studies of monomers and dimers containing 2'-fluoro-2'-deoxyadenosine. | Cheng, DM., et al. 1983. Biopolymers. 22: 1427-44. PMID: 6223669
- Synthesis and characterization of the dinucleoside monophosphates containing 2'-fluoro-2'-deoxyadenosine. | Uesugi, S., et al. 1981. Biochemistry. 20: 3056-62. PMID: 6972783
- Deoxyguanosine-resistant leukemia L1210 cells. Loss of specific deoxyribonucleoside kinase activity. | Cory, AH., et al. 1993. J Biol Chem. 268: 405-9. PMID: 8380161
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Fluoro-2′-deoxyadenosine, 250 mg | sc-225360 | 250 mg | $347.00 |
