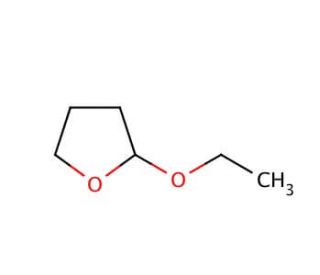
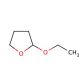
Molecular structure of 2-Ethoxytetrahydrofuran, CAS Number: 13436-46-9
2-Ethoxytetrahydrofuran (CAS 13436-46-9)
CAS Number:
13436-46-9
Molecular Weight:
116.16
Molecular Formula:
C6H12O2
Supplemental Information:
This is as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2-Ethoxytetrahydrofuran (2-ETHF) has garnered significant attention for its unique characteristics and versatility across various scientific domains. This colorless liquid, which easily dissolves in water, ether, and ethanol, is chiefly utilized as a solvent within the chemical sector. Its potential extends into biotechnology, where it plays a role in extracting DNA and RNA from biological specimens. Despite the incomplete understanding of its mechanism, the ability of 2-Ethoxytetrahydrofuran to form hydrogen bonds and its low viscosity are believed to enhance its solvent capabilities.
2-Ethoxytetrahydrofuran (CAS 13436-46-9) References
- Structure-activity relationships of 2,N(6),5'-substituted adenosine derivatives with potent activity at the A2B adenosine receptor. | Adachi, H., et al. 2007. J Med Chem. 50: 1810-27. PMID: 17378544
- Analysis of adducts in hepatic DNA of rats treated with N-nitrosopyrrolidine. | Wang, M., et al. 2007. Chem Res Toxicol. 20: 634-40. PMID: 17394361
- Brønsted acid catalyzed formal insertion of isocyanides into a C-O bond of acetals. | Tobisu, M., et al. 2007. J Am Chem Soc. 129: 11431-7. PMID: 17718490
- Recognition of cyclic, acyclic, exocyclic, and spiro acetals via structurally diagnostic ion/molecule reactions with the (CH3)2N-C(+)=O acylium ion. | Benassi, M., et al. 2008. J Org Chem. 73: 5549-57. PMID: 18570407
- Oxidative cleavage of diverse ethers by an extracellular fungal peroxygenase. | Kinne, M., et al. 2009. J Biol Chem. 284: 29343-9. PMID: 19713216
- Oxidation of cyclic acetals by ozone in ionic liquid media. | Van Doorslaer, C., et al. 2009. Chem Commun (Camb). 6439-41. PMID: 19841802
- A novel chemotype of kinase inhibitors: Discovery of 3,4-ring fused 7-azaindoles and deazapurines as potent JAK2 inhibitors. | Wang, T., et al. 2010. Bioorg Med Chem Lett. 20: 153-6. PMID: 19945871
- Ionic cyclodextrins in ionic liquid matrices as chiral stationary phases for gas chromatography. | Huang, K., et al. 2010. J Chromatogr A. 1217: 5261-73. PMID: 20609444
- Ion-molecule reactions: analytical and structural tool. | Osburn, S. and Ryzhov, V. 2013. Anal Chem. 85: 769-78. PMID: 23077968
- Total synthesis of aplykurodinone-1. | Liu, G., et al. 2014. Org Lett. 16: 4380-3. PMID: 25153682
- Hydrocarbon and phosphate triester formation during homolytic hydrolysis of organophosphonium ions: an alternate model for organophosphonate biodegradation. | Avila, LZ., et al. 1991. J Am Chem Soc. 113: 2242-6. PMID: 25906069
- Tryptophol and derivatives: natural occurrence and applications to the synthesis of bioactive compounds. | Palmieri, A. and Petrini, M. 2019. Nat Prod Rep. 36: 490-530. PMID: 30230504
- Scientific Opinion of Flavouring Group Evaluation 500 (FGE.500): rum ether. | , ., et al. 2017. EFSA J. 15: e04897. PMID: 32625610
- Studies on antitumor agents, 2. Syntheses and antitumor activities of 1-(tetrahydro-2-furanyl)-5-fluorouracil and 1,3-bis(tetrahydro-2-furanyl)-5-fluorouracil. | Yasumoto, M., et al. 1978. J Med Chem. 21: 738-41. PMID: 357721
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Ethoxytetrahydrofuran, 10 g | sc-225341 | 10 g | $561.00 |
