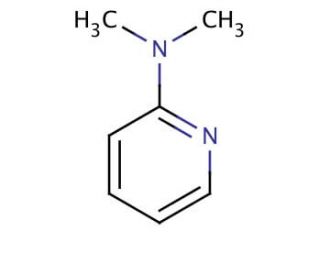
Molecular structure of 2-(Dimethylamino)pyridine, CAS Number: 5683-33-0
2-(Dimethylamino)pyridine (CAS 5683-33-0)
CAS Number:
5683-33-0
Molecular Weight:
122.17
Molecular Formula:
C7H10N2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2-(Dimethylamino)pyridine is a transparent liquid ranging from colorless to pale yellow. When combined with toluene at ambient temperature and in the company of 1.1 equivalents of methyl trifluoromethanesulfonate, it generates the (2-pyridyl)-trimethylammonium trifluoromethanesulfonate salt. This compound is used as a model substrate in crafting chelate carbenes through cyclometalation, H2 elimination, and reversible α-elimination.
2-(Dimethylamino)pyridine (CAS 5683-33-0) References
- Double geminal C-H activation and reversible alpha-elimination in 2-aminopyridine iridium(III) complexes: the role of hydrides and solvent in flattening the free energy surface. | Clot, E., et al. 2004. J Am Chem Soc. 126: 8795-804. PMID: 15250733
- Assessment of 4-(dimethylamino)pyridine as a capping agent for gold nanoparticles. | Gandubert, VJ. and Lennox, RB. 2005. Langmuir. 21: 6532-9. PMID: 15982063
- Iridium(III)-induced isomerization of 2-substituted pyridines to N-heterocyclic carbenes. | Alvarez, E., et al. 2006. J Am Chem Soc. 128: 13060-1. PMID: 17017779
- 2,2',2''-Terpyridine-catalyzed synthesis of cyclic carbonates from epoxides and carbon dioxide under solvent-free conditions. | Liu, H., et al. 2014. Int J Mol Sci. 15: 9945-51. PMID: 24901525
- Csp3 -H Activation without Chelation Assistance in an Iridium Pincer Complex Forming Cyclometallated Products. | Ahlstrand, DA., et al. 2017. Chemistry. 23: 1748-1751. PMID: 27982473
- Synthesis of 4-trifluoromethyl 2-pyrones and pyridones through the Brønsted base-catalyzed Pechmann-type reaction with cyclic 1,3-diones. | Yan, W., et al. 2018. Org Biomol Chem. 16: 9440-9445. PMID: 30515497
- Towards the enzymatic formation of artificial metal base pairs with a carboxy-imidazole-modified nucleotide. | Röthlisberger, P., et al. 2019. J Inorg Biochem. 191: 154-163. PMID: 30529723
- Preferential protonation and methylation at the nitrogen atoms of N, N‐dimethylamino derivatives of pyridine | Barbieri, G., Benassi, R., Grandi, R., Pagnoni, U. M., & Taddei, F. 1979. Organic Magnetic Resonance. 12(3): 159-162.
- Karramkam, M., Hinnen, F., Vaufrey, F., & Dollé, F. (2003). 2‐, 3‐and 4‐[18F] Fluoropyridine by no‐carrier‐added nucleophilic aromatic substitution with K [18F] F‐K222–a comparative study. | Karramkam, M., Hinnen, F., Vaufrey, F., & Dollé, F. 2003. Journal of Labelled Compounds and Radiopharmaceuticals: The Official Journal of the International Isotope Society. 46(10): 979-992.
- Synthesis and pH-dependent micellization of sulfonamide-modified diblock copolymer | Pal, R. R., Kim, M. S., & Lee, D. S. 2005. Macromolecular Research. 13: 467-476.
- Expedient Synthesis of (R)‐Curcuphenol: A Chiral Pool Strategy | Feng, J., Zhu, G., Liu, B., & Zhou, X. 2013. Chinese Journal of Chemistry. 31(1): 23-26.
- Pincer complexes, leading characters in C–H bond activation processes. Synthesis and catalytic applications | Valdés, H., Rufino-Felipe, E., & Morales-Morales, D. 2019. Journal of Organometallic Chemistry. 898: 120864.
- Preparation and characterization of modified starches obtained in acetic anhydride/tartaric acid medium | Tupa, M. V., Altuna, L., Herrera, M. L., & Foresti, M. L. 2020. Starch‐Stärke. 72(5-6): 1900300.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-(Dimethylamino)pyridine, 25 g | sc-237868 | 25 g | $91.00 |
