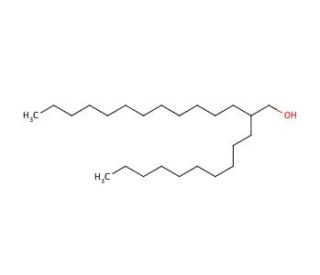
2-Decyl-1-tetradecanol (CAS 58670-89-6)
QUICK LINKS
2-Decyl-1-tetradecanol is a long-chain alcohol that functions as a surfactant in experimental applications. It acts by reducing the surface tension between two substances, allowing them to mix more easily. 2-Decyl-1-Tetradecanol′s mechanism of action involves its hydrophobic tail interacting with nonpolar substances, while its hydrophilic head interacts with polar substances, facilitating the formation of stable emulsions. In research and development, 2-Decyl-1-tetradecanol is utilized to aid in the dispersion of insoluble compounds in aqueous solutions, enabling the creation of stable formulations for experimental purposes. Its function as a surfactant allows for the manipulation of interfacial properties, which may be useful for experimental processes that require the controlled mixing of nonpolar and polar substances.
2-Decyl-1-tetradecanol (CAS 58670-89-6) References
- Design and synthesis of triglyceride analogue biotinylated suicide inhibitors for directed molecular evolution of lipolytic enzymes. | Deussen, HJ., et al. 2000. Bioorg Med Chem Lett. 10: 2027-31. PMID: 10987442
- Spin transition at the mesophase transition temperature in a cobalt(II) compound with branched alkyl chains. | Hayami, S., et al. 2007. Inorg Chem. 46: 7692-4. PMID: 17696425
- Specific surface modification of the acetylene-linked glycolipid vesicle by click chemistry. | Ito, H., et al. 2012. Chem Commun (Camb). 48: 5650-2. PMID: 22543823
- Advances in metal-free heterocycle-based columnar liquid crystals. | Roy, B., et al. 2012. Chemistry. 18: 14560-88. PMID: 23081719
- Dielectric relaxation of long-chain glass-forming monohydroxy alcohols. | Gao, Y., et al. 2013. J Chem Phys. 139: 164504. PMID: 24182046
- Production of Biomass-Based Automotive Lubricants by Reductive Etherification. | Jadhav, D., et al. 2017. ChemSusChem. 10: 2527-2533. PMID: 28406578
- Self-Assembly, Thermotropic, and Lyotropic Phase Behavior of Guerbet Branched-Chain Maltosides. | Saari, NAN., et al. 2018. Langmuir. 34: 8962-8974. PMID: 29999321
- Coexistence of two structural relaxation processes in monohydroxy alcohol-alkyl halogen mixtures: Dielectric and rheological studies. | Bierwirth, SP., et al. 2018. J Chem Phys. 149: 044509. PMID: 30068194
- Synthesis of Biomass-Derived Ethers for Use as Fuels and Lubricants. | Rorrer, JE., et al. 2019. ChemSusChem. 12: 2835-2858. PMID: 31232521
- Transforming Methyl Levulinate into Biosurfactants and Biolubricants by Chemoselective Reductive Etherification with Fatty Alcohols. | Garcia-Ortiz, A., et al. 2020. ChemSusChem. 13: 707-714. PMID: 31912979
- Evidence that Criegee intermediates drive autoxidation in unsaturated lipids. | Zeng, M., et al. 2020. Proc Natl Acad Sci U S A. 117: 4486-4490. PMID: 32071215
- Lipophilic Arginine Esters: The Gateway to Preservatives without Side Effects. | Shahzadi, I., et al. 2020. Mol Pharm. 17: 3129-3139. PMID: 32598849
- Experimental evidence that halogen bonding catalyzes the heterogeneous chlorination of alkenes in submicron liquid droplets. | Zeng, M. and Wilson, KR. 2021. Chem Sci. 12: 10455-10466. PMID: 34447538
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Decyl-1-tetradecanol, 100 ml | sc-230242 | 100 ml | $90.00 |
