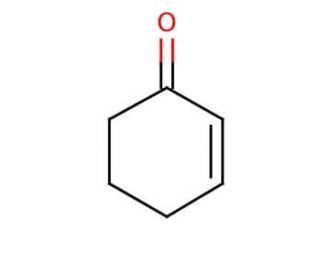
Molecular structure of 2-Cyclohexen-1-one, CAS Number: 930-68-7
2-Cyclohexen-1-one (CAS 930-68-7)
CAS Number:
930-68-7
Molecular Weight:
96.13
Molecular Formula:
C6H8O
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2-Cyclohexen-1-one is a chemical compound that functions as a reactant in various organic synthesis reactions. Its mechanism of action involves undergoing addition reactions with nucleophiles, such as Grignard reagents or organolithium compounds, to form new carbon-carbon bonds. This allows for the introduction of functional groups and the creation of complex molecular structures. 2-Cyclohexen-1-one can undergo reduction reactions to yield cyclohexanol, which can further serve as a building block in the synthesis of various organic compounds.
2-Cyclohexen-1-one (CAS 930-68-7) References
- Effects of glutathione depletion by 2-cyclohexen-1-one on excitatory amino acids-induced enhancement of activator protein-1 DNA binding in murine hippocampus. | Ogita, K., et al. 2001. J Neurochem. 76: 1905-15. PMID: 11259509
- Catalytic enantioselective conjugate addition of trimethylsilylacetylene to 2-cyclohexen-1-one. | Kwak, YS. and Corey, EJ. 2004. Org Lett. 6: 3385-8. PMID: 15355058
- Asymmetric Morita-Baylis-Hillman reaction of arylaldehydes with 2-cyclohexen-1-one catalyzed by chiral bis(thio)urea and DABCO. | Shi, M. and Liu, XG. 2008. Org Lett. 10: 1043-6. PMID: 18284244
- An exceptionally rapid and selective hydrogenation of 2-cyclohexen-1-one in supercritical carbon dioxide. | Chatterjee, M., et al. 2009. Chem Commun (Camb). 701-3. PMID: 19322427
- Participation of glucokinase inactivation in inhibition of glucose-induced insulin secretion by 2-cyclohexen-1-one. | Miwa, I., et al. 1990. Diabetes. 39: 1170-6. PMID: 2210070
- An alternative approach to para-C-H arylation of phenol: palladium-catalyzed tandem γ-arylation/aromatization of 2-cyclohexen-1-one derivatives. | Imahori, T., et al. 2012. Org Lett. 14: 1172-5. PMID: 22296212
- Influence of Processing Conditions on the Flavor Profiles of Mulberry (Morus alba Linn) Fruits Using Instrumental Flavor Analysis and Descriptive Sensory Analysis. | Hwang, IS. and Kim, MK. 2020. Foods. 9: PMID: 32380639
- An integrated giant polyoxometalate complex for photothermally enhanced catalytic oxidation. | Chen, X., et al. 2021. Sci Adv. 7: PMID: 34301598
- C-H Bond Activation Mechanism by a Pd(II)-(μ-O)-Au(0) Structure Unique to Heterogeneous Catalysts. | Takei, D., et al. 2022. JACS Au. 2: 394-406. PMID: 35252989
- Consecutive reactions to construct tricarbonyl compounds and synthetic applications thereof. | Madroñero, D., et al. 2021. RSC Adv. 11: 33235-33244. PMID: 35497560
- Immobilization of Ene Reductase in Polyvinyl Alcohol Hydrogel. | Alagöz, D., et al. 2022. Protein J. 41: 394-402. PMID: 35715719
- Changes of volatile substance composition during processing of nine-processed tangerine peel (Jiuzhi Chenpi) determined by gas chromatography-ion mobility spectrometry. | Fu, M., et al. 2022. Front Nutr. 9: 963655. PMID: 36091238
- Differentiating Huangjiu with Varying Sugar Contents from Different Regions Based on Targeted Metabolomics Analyses of Volatile Carbonyl Compounds. | Yu, J., et al. 2023. Foods. 12: PMID: 37048277
- Artefenomel Regioisomer RLA-3107 Is a Promising Lead for the Discovery of Next-Generation Endoperoxide Antimalarials. | Blank, BR., et al. 2023. ACS Med Chem Lett. 14: 493-498. PMID: 37077383
- Progress on the Cu-Catalyzed 1,4-Conjugate Addition to Thiochromones. | Guo, F., et al. 2023. Catalysts. 13: PMID: 37293477
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Cyclohexen-1-one, 10 ml | sc-254227 | 10 ml | $27.00 | |||
2-Cyclohexen-1-one, 25 ml | sc-254227A | 25 ml | $37.00 |
