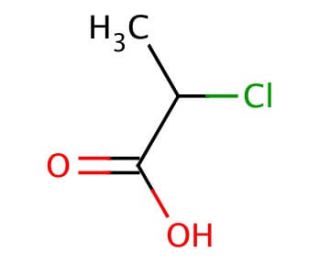
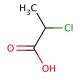
2-Chloropropionic acid (CAS 598-78-7)
QUICK LINKS
2-Chloropropionic acid, a chlorinated carboxylic acid, stands as a pivotal compound in synthetic organic chemistry and research, primarily due to its role as a reagent in the synthesis of other complex molecules. Its mechanism of action hinges on its reactivity as a halogenated acid, which enables it to participate in various chemical reactions, such as nucleophilic substitution reactions, where it can donate its chlorine atom to other molecules, thereby acting as an alkylating agent. This property is particularly valuable in the construction of more complex organic compounds, including the synthesis of esters, amides, and other derivatives, making it an essential tool in the development of new materials and chemicals. Additionally, its involvement in the production of herbicides and biocides highlights its broader significance in agricultural research and pest management strategies. The versatility and reactivity of 2-Chloropropionic acid thus contribute substantially to its prominence in chemical research, facilitating advancements in synthetic methodologies and the exploration of novel compounds with potential applications in various scientific fields.
2-Chloropropionic acid (CAS 598-78-7) References
- Symposium overview: the role of glutathione in neuroprotection and neurotoxicity. | Monks, TJ., et al. 1999. Toxicol Sci. 51: 161-77. PMID: 10543018
- Cerebellum as a target for toxic substances. | Fonnum, F. and Lock, EA. 2000. Toxicol Lett. 112-113: 9-16. PMID: 10720707
- Photoreduction of the chloropropionic acid of carfentrazone-ethyl in sodium sulfide [corrected]. | Ngim, KK. and Crosby, DG. 2002. Environ Toxicol Chem. 21: 2007-13. PMID: 12371474
- Putrescine as a marker of the effects of 2-chloropropionic acid in the rat brain. | de Vera, N., et al. 2004. Neurosci Lett. 362: 209-12. PMID: 15158016
- Effects of strong and weak hydrogen bond formation on VCD spectra: a case study of 2-chloropropionic acid. | Góbi, S., et al. 2011. Phys Chem Chem Phys. 13: 13972-84. PMID: 21701707
- Near-infrared radiation induced conformational change and hydrogen atom tunneling of 2-chloropropionic acid in low-temperature Ar matrix. | Bazsó, G., et al. 2012. J Phys Chem A. 116: 4823-32. PMID: 22554056
- Improvement of NADPH bioavailability in Escherichia coli through the use of phosphofructokinase deficient strains. | Wang, Y., et al. 2013. Appl Microbiol Biotechnol. 97: 6883-93. PMID: 23558585
- Theoretical analyses on enantiospecificity of L-2-haloacid dehalogenase (DehL) from Rhizobium sp. RC1 towards 2-chloropropionic acid. | Adamu, A., et al. 2019. J Mol Graph Model. 92: 131-139. PMID: 31352207
- Different Types of Ligand Exchange Induced by Au Substitution in a Maintained Nanocluster Template. | Du, W., et al. 2020. Inorg Chem. 59: 1675-1681. PMID: 31944677
- In Silico Analysis on the Interaction of Haloacid Dehalogenase from Bacillus cereus IndB1 with 2-Chloroalkanoic Acid Substrates. | Ratnaningsih, E. and Saepulloh, S. 2022. ScientificWorldJournal. 2022: 1579194. PMID: 36254337
- Cloning, sequencing and expression in Escherichia coli of two Rhizobium sp. genes encoding haloalkanoate dehalogenases of opposite stereospecificity. | Cairns, SS., et al. 1996. Eur J Biochem. 235: 744-9. PMID: 8654424
- Neuropathological changes in rat brain following oral administration of 2-chloropropionic acid. | Simpson, MG., et al. 1996. Neurotoxicology. 17: 471-80. PMID: 8856742
- Haloalkanoate dehalogenase II (DehE) of a Rhizobium sp.--molecular analysis of the gene and formation of carbon monoxide from trihaloacetate by the enzyme. | Stringfellow, JM., et al. 1997. Eur J Biochem. 250: 789-93. PMID: 9461303
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Chloropropionic acid, 100 g | sc-256123 | 100 g | $22.00 |
