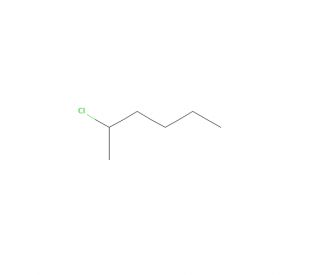
2-Chlorohexane (CAS 638-28-8)
QUICK LINKS
2-Chlorohexane is a colorless, highly volatile liquid characterized by a potent odor. Being a chlorinated hydrocarbon, it serves as an essential intermediary in the synthesis of various chemicals and plastics. Moreover, it finds utility as a solvent in the manufacturing of pharmaceuticals, paints, and coatings. It is vital to exercise extreme caution when handling 2-Chlorohexane due to its highly toxic and flammable nature. The mechanism of action of this chlorinated hydrocarbon stems from its ability to undergo reactions with other molecules. Notably, it can react with hydrocarbons, alcohols, and amines, resulting in the formation of diverse products. These reactions involving 2-Chlorohexane are typically exothermic in nature.
2-Chlorohexane (CAS 638-28-8) References
- Selective functionalization of C(sp3)-H bonds: catalytic chlorination and bromination by IronIII-acacen-halide under ambient condition. | Shen, C., et al. 2022. Chem Commun (Camb). 58: 10627-10630. PMID: 36069398
- The Peroxide-and Light-Induced Reactions of Trichloromethanesulfonyl Chloride with Hydrocarbons1 | Huyser, E. S., & Giddings, B. 1962. The Journal of Organic Chemistry. 27(10): 3391-3395.
- Condensation of. alpha.-olefins with paraformaldehyde, acetylating agents, and hydrogen chloride | Stapp, P. R. 1971. The Journal of Organic Chemistry. 36(17): 2505-2507.
- Ionic chlorination of alkanes with sulfuryl chloride-sulfolane | Tabushi, I., Yoshida, Z., & Tamaru, Y. 1973. Tetrahedron. 29(1): 81-84.
- Free radical substitution in aliphatic compounds. Part 33. Halogen atom abstraction from alkyl halides by trimethylgermanium radicals in the gas phase | Coates, D. A., & Tedder, J. M. 1978. Journal of the Chemical Society, Perkin Transactions 2. (8): 725-728.
- Cyclic chlorination reactions induced by visible light with chlorocopper (II) complexes as mediators | Cervone, E., & Camassei, F. D. 1981. Journal of Photochemistry. 15(3): 203-212.
- Carbon‐13 nuclear magnetic resonance of mono‐and di‐chloro‐hexanes and mono‐and di‐chloro‐heptanes. Assignment of configurations | Nouguier, R., Surzur, J. M., & Virgili, A. 1981. Organic Magnetic Resonance. 15(2): 155-157.
- A macromolecular conformational change driven by a minute chiral solvation energy | Green, M. M., Khatri, C., & Peterson, N. C. 1993. Journal of the American Chemical Society. 115(11): 4941-4942.
- Mechanism of Photoinduced Isomerization of Alkyl Radicals Trapped in 77 K Solids | Koizumi, H., Kosugi, S., & Yoshida, H. 1996. The Journal of Physical Chemistry. 100(12): 4848-4852.
- Effects of alkyl chain structure on carbon− halogen bond dissociation and β-hydride elimination by alkyl halides on a Cu (100) surface | Lin, J. L., Teplyakov, A. V., & Bent, B. E. 1996. The Journal of Physical Chemistry. 100(25): 10721-10731.
- Optimization of the Penicillin Ring Expansion Reaction through the Use of an Alkene as an HCl Scavenger | Copp, J. D., & Tharp, G. A. 1997. Organic Process Research & Development. 1(1): 92-94.
- Effect of molecular packing on radiolytic processes of n-alkanes | Ceulemans, J. 2003. Radiation physics and chemistry. 67(3-4): 199-205.
- Overview and assessment of recent research on the structural defects in poly (vinyl chloride) | Starnes Jr, W. H. 2012. Polymer degradation and stability. 97(9): 1815-1821.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Chlorohexane, 5 g | sc-486954 | 5 g | $204.00 |
