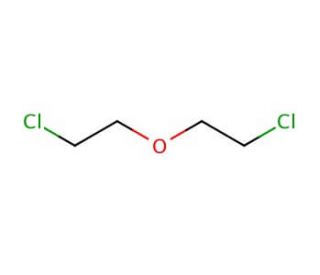
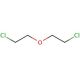
2-Chloroethyl ether (CAS 111-44-4)
QUICK LINKS
2-Chloroethyl ether is extensively used in research, particularly in the field of organic synthesis, where it acts as a versatile alkylating agent. 2-Chloroethyl Ether is for introducing the 2-chloroethyl group into other chemical structures, thereby enabling the study of molecular interactions and reaction mechanisms that are relevant to the development of new materials and chemicals. 2-Chloroethyl ether is investigated for its potential in synthesizing ethers and esters, which are important in various industrial applications such as solvents and intermediates in the production of more complex molecules. The study of 2-Chloroethyl ether′s reactivity and stability under different conditions is also critical for ensuring safe handling and effective application in both laboratory and industrial applications.
2-Chloroethyl ether (CAS 111-44-4) References
- Bis(2-chloroethyl)ether. | . 1999. IARC Monogr Eval Carcinog Risks Hum. 71 Pt 3: 1265-9. PMID: 10476394
- Sulfur mustard-stimulated protease: a target for antivesicant drugs. | Ray, P., et al. 2002. J Appl Toxicol. 22: 139-40. PMID: 11920939
- Degradation of 2-Chloroethylvinylether by Ancylobacter aquaticus AD25 and AD27. | van den Wijngaard, AJ., et al. 1993. Appl Environ Microbiol. 59: 2777-83. PMID: 16349032
- The discovery of crown ethers. | Pedersen, CJ. 1988. Science. 241: 536-40. PMID: 17774576
- Biodegradation of bis(2-chloroethyl) ether by Xanthobacter sp. strain ENV481. | McClay, K., et al. 2007. Appl Environ Microbiol. 73: 6870-5. PMID: 17873075
- Surface catalyzed Fenton treatment of bis(2-chloroethyl) ether and bis(2-chloroethoxy) methane. | Mutuc, MD., et al. 2008. Chemosphere. 70: 1390-8. PMID: 18061235
- Mobility and sorption of bis-2-chloroethyl ether in an aquifer material. | Bednar, AJ., et al. 2009. J Hazard Mater. 168: 1041-6. PMID: 19345490
- Chemical oxidation of bis(2-chloroethyl) ether in the Fenton process: Kinetics, pathways and toxicity assessment. | Shi, J., et al. 2017. Chemosphere. 180: 117-124. PMID: 28395149
- Assessment of anaerobic biodegradation of bis(2-chloroethyl) ether in groundwater using carbon and chlorine compound-specific isotope analysis. | Segal, DC., et al. 2018. Sci Total Environ. 625: 696-705. PMID: 29306157
- Mechanistic investigation of the atmospheric oxidation of bis(2-chloroethyl) ether (ClCH2CH2OCH2CH2Cl) by OH and NO3 radicals and Cl atoms: a DFT approach. | Paul, S., et al. 2019. J Mol Model. 25: 43. PMID: 30675641
- Characteristics and Mechanism of Vinyl Ether Cationic Polymerization in Aqueous Media Initiated by Alcohol/B(C₆F₅)₃/Et₂O. | Zhang, J., et al. 2019. Polymers (Basel). 11: PMID: 30960484
- Degradation kinetics and mechanism of bis(2-chloroethyl) ether by electromagnetic induction electrodeless lamp activated persulfate. | Zhou, Y., et al. 2020. Chemosphere. 261: 127709. PMID: 32745742
- Thiodiglycolic acid: a major metabolite of bis(2-chloroethyl)ether. | Lingg, RD., et al. 1979. Toxicol Appl Pharmacol. 47: 23-34. PMID: 425117
- Metabolism of bis(2-chloroethyl)ether and bis(2-chloroisopropyl)ether in the rat. | Lingg, RD., et al. 1982. Arch Environ Contam Toxicol. 11: 173-83. PMID: 6807217
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Chloroethyl ether, 5 g | sc-238000 | 5 g | $32.00 | |||
2-Chloroethyl ether, 100 g | sc-238000A | 100 g | $45.00 |
