2-Chlorobenzaldehyde (CAS 89-98-5)
QUICK LINKS
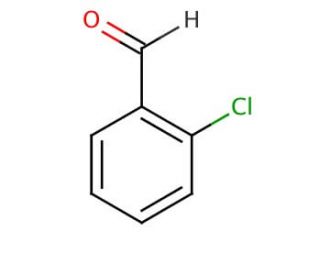
2-Chlorobenzaldehyde, also known as 2-CBA, is an aromatic aldehyde characterized by the molecular formula C7H5ClO. Its appearance is that of a colorless liquid, and it possesses a distinct odor. This compound plays a role as a precursor in the synthesis of various significant compounds. Its versatile applications encompass the synthesis of fragrances, and dyes, along with its contribution to the production of polymers, plasticizers, and surfactants. Scientific research has extensively explored the properties of 2-Chlorobenzaldehyde. It serves as a model compound for studying the reactivity of aldehydes and the intricacies of the Claisen-Schmidt reaction. Moreover, the compound has repeatedly proved its worth in various drug, fragrance, and dye synthesis processes, while also proving valuable in the creation of polymers, plasticizers, and surfactants. As an aldehyde, 2-Chlorobenzaldehyde exhibits a range of chemical reactions, notably including aldol condensation, Claisen-Schmidt reaction, and Cannizzaro reaction. During aldol condensation, it reacts with an alcohol to yield an aldol product. The Claisen-Schmidt reaction, on the other hand, involves its reaction with a ketone, resulting in the formation of a β-keto-aldehyde. Lastly, the Cannizzaro reaction entails the reaction of 2-Chlorobenzaldehyde with an alkali to generate an alcohol. 2-Chlorobenzaldehyde stands as a vital compound in the realm of chemical synthesis, showcasing its value in numerous applications and its role as a subject of extensive scientific investigation.
2-Chlorobenzaldehyde (CAS 89-98-5) References
- Preliminary characterization of four 2-chlorobenzoate-degrading anaerobic bacterial consortia. | Genthner, BR. 1999. Biodegradation. 10: 27-33. PMID: 10423838
- Synthesis, Crystal Structure, and Second-Order Optical Nonlinearity of Bis(2-chlorobenzaldehyde thiosemicarbazone)cadmium Halides (CdL(2)X(2); X = Br, I). | Tian Yp, YP., et al. 1997. Inorg Chem. 36: 1247-1252. PMID: 11669693
- SYNTHESIS OF SOME DIAMIDES OF 2-CHLOROBENZALDEHYDE, 2,4-DICHLOROBENZALDEHYDE, AND 3,4-DICHLOROBENZALDEHYDE. | LAROCCA, JP. and CULPEPPER, WC. 1965. J Pharm Sci. 54: 489-90. PMID: 14301600
- Synthesis of 1-aryl-1H-indazoles via palladium-catalyzed intramolecular amination of aryl halides. | Lebedev, AY., et al. 2005. J Org Chem. 70: 596-602. PMID: 15651807
- Vibrational spectra of partially oriented molecules having two conformers in nematic and isotropic solutions: furfural and 2-chlorobenzaldehyde. | Rogojerov, M., et al. 2005. Spectrochim Acta A Mol Biomol Spectrosc. 61: 1661-70. PMID: 15820900
- Investigation of catalytic characterization of two-dimensional molecular space with regular ammonium and pyridine groups. | Chen, J., et al. 2009. Langmuir. 25: 5993-9. PMID: 19296641
- Imidazo[1,2-a]pyridin-3-amines as potential HIV-1 non-nucleoside reverse transcriptase inhibitors. | Bode, ML., et al. 2011. Bioorg Med Chem. 19: 4227-37. PMID: 21700466
- Fast and catalyst-free hydrazone ligation via ortho-halo-substituted benzaldehydes for protein C-terminal labeling at neutral pH. | Xu, Y., et al. 2015. Chem Commun (Camb). 51: 13189-13192. PMID: 26195073
- SYNTHESIS AND ANTIMICROBIAL ACTIVITY OF 5-SUBSTITUED 4-THIAZOLIDINONES WITH SULFANILAMIDE PHARMACOPHORE. | Zevzikoviene, A., et al. 2016. Acta Pol Pharm. 73: 1155-1161. PMID: 29638056
- Percutaneous absorption of 14C-labelled 2-chlorobenzaldehyde in rats. Metabolism and toxicokinetics. | Rietveld, EC., et al. 1988. Eur J Drug Metab Pharmacokinet. 13: 231-40. PMID: 3243317
- Synthesis, anticancer evaluation and molecular docking studies of methotrexate's novel Schiff base derivatives against malignant glioma cell lines. | Nemat, A., et al. 2022. J Biomol Struct Dyn. 40: 2865-2877. PMID: 33183168
- Soft threshold partial least squares predicts the survival fraction of malignant glioma cells against different concentrations of methotrexate's derivatives. | Mehmood, T. and Iqbal, M. 2021. Sci Rep. 11: 18741. PMID: 34548518
- Synthesis and Aminomethylation of 2-Amino-4-(2-chlorophenyl)-6-(dicyanomethyl)-1,4-dihydropyridine-3,5-dicarbonitrile N-Methylmorpholinium Salt. | Kurskova, AO., et al. 2022. Russ J Gen Chem. 92: 779-790. PMID: 35729943
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Chlorobenzaldehyde, 100 g | sc-237991 | 100 g | $30.00 | |||
2-Chlorobenzaldehyde, 500 g | sc-237991A | 500 g | $58.00 |
