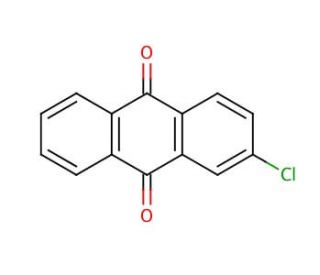
2-Chloroanthraquinone (CAS 131-09-9)
See product citations (1)
QUICK LINKS
2-Chloroanthraquinone is a chemical compound that functions as an intermediate in the synthesis of dyes and pigments. It acts as an electron acceptor in organic synthesis, participating in various reactions such as Friedel-Crafts acylation and nucleophilic substitution. 2-Chloroanthraquinone′s mode of action involves its ability to undergo electrophilic aromatic substitution, allowing it to react with nucleophiles to form new carbon-carbon or carbon-heteroatom bonds. 2-Chloroanthraquinone is utilized as a building block for the preparation of complex organic molecules, enabling the creation of diverse chemical structures with potential applications in various fields. Its reactivity and selectivity make it useful for the modification of organic compounds, contributing to the advancement of methodologies and the discovery of new chemical entities.
2-Chloroanthraquinone (CAS 131-09-9) References
- Inhibition of sulfate respiration by 1,8-dihydroxyanthraquinone and other anthraquinone derivatives. | Cooling, FI., et al. 1996. Appl Environ Microbiol. 62: 2999-3004. PMID: 16535385
- Hybridization properties of deoxyoligonucleotides containing anthraquinone pseudonucleosides. | Lin, KY. and Matteucci, M. 1991. Nucleic Acids Res. 19: 3111-4. PMID: 1711680
- Organocatalytic oxidative dehydrogenation of dihydroarenes by dioxygen using 2,3-dichloro-5,6-dicyano-benzoquinone (DDQ) and NaNO2. | Zhang, W., et al. 2008. Molecules. 13: 3236-45. PMID: 19104488
- Alkylation of Nonacidic C(sp3)-H Bonds by Photoinduced Catalytic Michael-Type Radical Addition. | Kamijo, S., et al. 2016. Org Lett. 18: 4912-4915. PMID: 27668684
- Hydrogen Atom Transfer (HAT): A Versatile Strategy for Substrate Activation in Photocatalyzed Organic Synthesis. | Capaldo, L. and Ravelli, D. 2017. European J Org Chem. 2017: 2056-2071. PMID: 30147436
- Identification and Characterization of a Novel N- and O-Glycosyltransferase from Saccharopolyspora erythraea. | Gutacker, F., et al. 2020. Molecules. 25: PMID: 32727097
- Standards Development For Differential Scanning Calorimetry. | Callanan, JE., et al. 1986. J Res Natl Bur Stand (1977). 91: 123-129. PMID: 34345075
- Direct Photocatalyzed Hydrogen Atom Transfer (HAT) for Aliphatic C-H Bonds Elaboration. | Capaldo, L., et al. 2022. Chem Rev. 122: 1875-1924. PMID: 34355884
- Site-Selective Acceptorless Dehydrogenation of Aliphatics Enabled by Organophotoredox/Cobalt Dual Catalysis. | Zhou, MJ., et al. 2021. J Am Chem Soc. 143: 16470-16485. PMID: 34592106
- Metal-free photoredox-catalyzed direct α-oxygenation of N,N-dibenzylanilines to imides under visible light. | Neerathilingam, N. and Anandhan, R. 2022. RSC Adv. 12: 8368-8373. PMID: 35424823
- Trade-Off between Redox Potential and the Strength of Electrochemical CO2 Capture in Quinones. | Bui, AT., et al. 2022. J Phys Chem C Nanomater Interfaces. 126: 14163-14172. PMID: 36051254
- External Catalyst- and Additive-Free Photo-Oxidation of Aromatic Alcohols to Carboxylic Acids or Ketones Using Air/O2. | Xu, M., et al. 2023. Molecules. 28: PMID: 37049794
- Ionic Liquid-Supported Photocatalysts: A Reusable Environmentally Friendly Oxidation Reaction System That Uses Air and Light. | Koguchi, S., et al. 2023. Int J Mol Sci. 24: PMID: 37108301
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Chloroanthraquinone, 25 g | sc-225308 | 25 g | $70.00 | |||
2-Chloroanthraquinone, 250 g | sc-225308A | 250 g | $93.00 |
