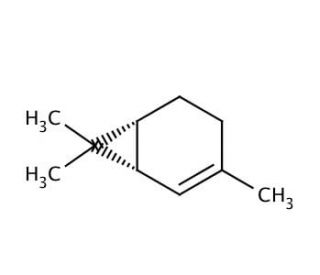
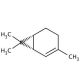
(+)-2-Carene (CAS 4497-92-1)
QUICK LINKS
(+)-2-Carene is a naturally occurring bicyclic monoterpene that is of interest in the field of organic chemistry and materials science. It is studied for its potential as a starting material in the synthesis of terpenoids and other complex organic molecules. The structure of (+)-2-Carene, featuring a strained bicyclic system, is relevant to research focusing on strain-release mechanisms during chemical reactions. In addition, its reactivity is exploited in the study of rearrangement reactions, polymerization processes, and the development of novel organic synthesis strategies. Researchers also investigate the compound for its ability to serve as a chiral building block, which can introduce stereochemical complexity into synthetic compounds. The applications of (+)-2-Carene extend to the field of materials science, where it is examined for its potential use in the development of renewable resources for the production of resins and polymers.
(+)-2-Carene (CAS 4497-92-1) References
- Response factor considerations for the quantitative analysis of western redcedar (Thuja plicata) foliar monoterpenes. | Kimball, BA., et al. 2005. J Chromatogr Sci. 43: 253-8. PMID: 15975244
- Lippia javanica (Burm F) Spreng: its general constituents and bioactivity on mosquitoes. | Nzira, L., et al. 2009. Trop Biomed. 26: 85-91. PMID: 19696732
- Biotransformation of (1S)-2-carene and (1S)-3-carene by Picea abies suspension culture. | Dvorakova, M., et al. 2011. Molecules. 16: 10541-55. PMID: 22183881
- A tale of two carenes: intrinsic optical activity and large-amplitude nuclear displacement. | Lahiri, P., et al. 2012. J Phys Chem A. 116: 9516-33. PMID: 22888793
- Conformational Effects on Specific Rotation: A Theoretical Study Based on the S̃k Method. | Caricato, M. 2015. J Phys Chem A. 119: 8303-10. PMID: 26167864
- 7-Step total synthesis of (+)-EBC-329: Photoisomerisation reveals new seco-casbane family member. | Vanden Berg, TJ., et al. 2017. Org Biomol Chem. 15: 7102-7105. PMID: 28820535
- Design, Synthesis, and Biological Investigation of Novel Classes of 3-Carene-Derived Potent Inhibitors of TDP1. | Il'ina, IV., et al. 2020. Molecules. 25: PMID: 32751997
- Compounds Associated with Infection by the Root-Knot Nematode, Meloidogyne javanica, Influence the Ability of Infective Juveniles to Recognize Host Plants. | Kihika, R., et al. 2020. J Agric Food Chem. 68: 9100-9109. PMID: 32786872
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(+)-2-Carene, 5 ml | sc-237964 | 5 ml | $335.00 |
