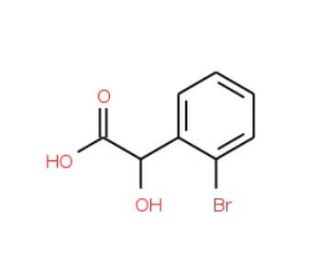
2-Bromomandelic acid (CAS 7157-15-5)
QUICK LINKS
2-Bromomandelic acid, referred to as 2-BPA, is an organic compound classified as a phenylcarboxylic acid. This compound manifests as a white crystalline solid with a molecular weight of 247.02 g/mol and a melting point ranging from 93 to 95° C. Furthermore, it has facilitated the exploration of enzyme mechanisms, shedding light on their intricate workings. The compound has also proven valuable in organic compound synthesis and the investigation of chemical reactions. Additionally, 2-Bromomandelic acid has played a significant role in the synthesis of metal complexes, enabling the study of their properties and interactions. Moreover, it has been instrumental in examining electrochemical processes, broadening the scope of scientific understanding. In terms of its mechanism of action, 2-Bromomandelic acid has demonstrated its efficacy as an enzyme inhibitor, particularly targeting enzymes involved in drug metabolism. Specifically, it inhibits the activity of cytochrome P450 2C9 (CYP2C9), a enzyme responsible for metabolizing drugs such as warfarin, phenytoin, and tolbutamide. Additionally, the compound has been observed to inhibit cytochrome P450 3A4 (CYP3A4), another vital enzyme involved in drug metabolism. Notably, drugs such as tamoxifen, cyclosporine, and carbamazepine rely on CYP3A4 for their breakdown and elimination.
2-Bromomandelic acid (CAS 7157-15-5) References
- Preparation of a β-cyclodextrin functionalized monolith via a novel and simple one-pot approach and application to enantioseparations. | Zhang, Q., et al. 2014. J Chromatogr A. 1325: 147-54. PMID: 24377739
- Synthetic studies of 7-oxygenated aporphine alkaloids: preparation of (-)-oliveroline, (-)-nornuciferidine, and derivatives. | Ku, AF. and Cuny, GD. 2015. Org Lett. 17: 1134-7. PMID: 25710592
- Enantiomeric NMR discrimination of carboxylic acids using actinomycin D as a chiral solvating agent. | Bai, L., et al. 2019. Org Biomol Chem. 17: 1466-1470. PMID: 30672950
- Construction of β-Cyclodextrin Covalent Organic Framework-Modified Chiral Stationary Phase for Chiral Separation. | Wang, Y., et al. 2019. ACS Appl Mater Interfaces. 11: 48363-48369. PMID: 31794183
- Discrimination in resolving systems. II: Ephedrine-substituted mandelic acids. | Valente, EJ., et al. 1995. Chirality. 7: 652-76. PMID: 8593258
- Relationships between the racemic structures of substituted mandelic acids containing 8- and 10-membered hydrogen bonded dimer rings† | S. J. Coles, ORCID logo *a A. L. Ellis,a K. Leung,a J. Sarson,a T. L. Threlfalla and G. J. Tizzarda. 2014. CrystEngComm,.,16,: 10816-10823.
- Solid Phase Behavior in the Chiral Systems of Various 2-Hydroxy-2-phenylacetic Acid (Mandelic Acid) Derivatives | Jan von Langermann*†‡, Erik Temmel‡, Andreas Seidel-Morgenstern‡§, and Heike Lorenz*‡. 2015. J. Chem. Eng. Data., 60, 3,: 721–728.
- Chemical Insights from Systematic Structural Studies. The 'Stamp Collecting' Approach to Understanding the Solid State | , et al. 30 June 2016. Crystallizing Ideas – The Role of Chemistry. pp 1–21.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Bromomandelic acid, 1 g | sc-307243 | 1 g | $200.00 |
