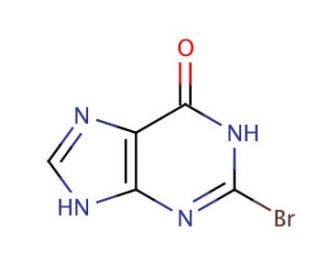
2-Bromohypoxanthine (CAS 87781-93-9)
QUICK LINKS
2-Bromohypoxanthine is a compound utilized in biochemical research focused on nucleic acid chemistry and enzyme-substrate interactions. It serves as an analog of hypoxanthine, a naturally occurring purine derivative, to study the effects of halogenation on base pairing and nucleic acid stability. Investigations involving 2-Bromohypoxanthine are relevant to understanding the mutagenic potential of brominated nucleobases. Additionally, this compound is used in the study of purine salvage pathways and the enzymes involved in nucleotide metabolism. It can also be employed in the design of molecular probes and in the development of methods for detecting specific nucleic acid sequences. The reactivity of 2-Bromohypoxanthine with various nucleophiles is of particular interest in synthetic organic chemistry for the generation of modified nucleotides.
2-Bromohypoxanthine (CAS 87781-93-9) References
- Resin-capture and release strategy toward combinatorial libraries of 2,6,9-substituted purines. | Ding, S., et al. 2002. J Comb Chem. 4: 183-6. PMID: 11886295
- Inhibition of herpes simplex virus thymidine kinases by 2-phenylamino-6-oxopurines and related compounds: structure-activity relationships and antiherpetic activity in vivo. | Manikowski, A., et al. 2005. J Med Chem. 48: 3919-29. PMID: 15916444
- Design and antimicrobial action of purine analogues that bind Guanine riboswitches. | Kim, JN., et al. 2009. ACS Chem Biol. 4: 915-27. PMID: 19739679
- Structure-activity relationships of N2-substituted guanines as inhibitors of HSV1 and HSV2 thymidine kinases. | Hildebrand, C., et al. 1990. J Med Chem. 33: 203-6. PMID: 2153203
- Formation of N2,3-ethanoguanine in DNA after in vitro treatment with the therapeutic agent, N-(2-chloroethyl)-N'-cyclohexyl-N-nitrosourea. | Habraken, Y., et al. 1990. Carcinogenesis. 11: 223-8. PMID: 2302749
- Supercritical fluid extraction as a preparation method for mass spectrometry of dried blood spots. | Matsubara, A., et al. 2014. J Chromatogr B Analyt Technol Biomed Life Sci. 969: 199-204. PMID: 25178194
- Synthesis and biological properties of purine and pyrimidine 5'-deoxy-5'-(dihydroxyphosphinyl)-beta-D-ribofuranosyl analogues of AMP, GMP, IMP, and CMP. | Raju, N., et al. 1989. J Med Chem. 32: 1307-13. PMID: 2542559
- Noncovalent and site-directed spin labeling of duplex RNA. | Kamble, NR., et al. 2016. Chem Commun (Camb). 52: 14442-14445. PMID: 27901530
- Evaluation of a novel system for analyzing hydrophilic blood metabolites. | Nishiumi, S., et al. 2017. J Biosci Bioeng. 123: 754-759. PMID: 28291661
- Use of on-line supercritical fluid extraction-supercritical fluid chromatography/tandem mass spectrometry to analyze disease biomarkers in dried serum spots compared with serum analysis using liquid chromatography/tandem mass spectrometry. | Suzuki, M., et al. 2017. Rapid Commun Mass Spectrom. 31: 886-894. PMID: 28332299
- Differences in metabolite profiles caused by pre-analytical blood processing procedures. | Nishiumi, S., et al. 2018. J Biosci Bioeng. 125: 613-618. PMID: 29258730
- Synthesis and characterization of N2-(p-n-butylphenyl)-2'-deoxyguanosine and its 5'-triphosphate and their inhibition of HeLa DNA polymerase alpha. | Wright, GE. and Dudycz, LW. 1984. J Med Chem. 27: 175-81. PMID: 6694166
- N2-amination of guanine to 2-hydrazinohypoxanthine, a novel in vivo nucleic acid modification produced by the hepatocarcinogen 2-nitropropane. | Sodum, RS. and Fiala, ES. 1998. Chem Res Toxicol. 11: 1453-9. PMID: 9860487
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Bromohypoxanthine, 1 g | sc-251715 | 1 g | $224.00 |
