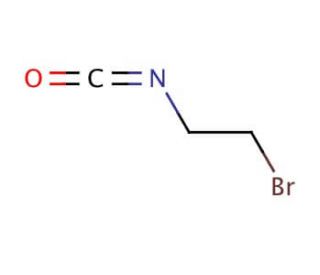
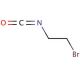
2-Bromoethyl isocyanate (CAS 42865-19-0)
QUICK LINKS
2-Bromoethyl isocyanate, an electrophilic compound, possesses unique properties that make it highly valuable in scientific research. This colorless liquid exhibits a pungent odor and remarkable reactivity. Its applications span various research fields, including organic synthesis, medicinal chemistry, and material science. This compound serves as a versatile building block in organic synthesis, enabling the creation of diverse compounds. In medicinal chemistry, it contributes to the development of novel drugs and pharmaceuticals. Additionally, it finds utility in material science, where it can modify the properties of polymers and other materials. While the precise mechanism of action for 2-Bromoethyl isocyanate remains elusive, it is postulated to engage in reactions with nucleophilic groups within proteins and other biomolecules, resulting in the formation of covalent bonds. This, in turn, can induce modifications in the structure and function of these biomolecules, yielding a wide array of effects. In summary, 2-Bromoethyl isocyanate stands as a compound within scientific research due to its electrophilic nature and broad range of applications. Its usage spans organic synthesis, medicinal chemistry, and material science, with potential for significant impact in understanding and manipulating biomolecular systems.
2-Bromoethyl isocyanate (CAS 42865-19-0) References
- Quaternary ammonium functionalized poly(propylene imine) dendrimers as effective antimicrobials: structure-activity studies. | Chen, CZ., et al. 2000. Biomacromolecules. 1: 473-80. PMID: 11710139
- Synthesis, DNA-binding activity and cytotoxicity of carbamate derivatives of Hoechst 33258 in breast cancer MCF-7 cells. | Bielawski, K., et al. 2002. Biol Pharm Bull. 25: 916-9. PMID: 12132669
- Halogenoalkyl isocyanates as bifunctional reagents in an Aza-Wittig/heterocyclization reaction on the solid phase: efficient entry into new tetracyclic benzimidazole systems. | Hoesl, CE., et al. 2004. J Comb Chem. 6: 220-3. PMID: 15002970
- A simple approach to sensor discovery and fabrication on self-assembled monolayers on glass. | Basabe-Desmonts, L., et al. 2004. J Am Chem Soc. 126: 7293-9. PMID: 15186166
- Chloride selective calix[4]arene optical sensor combining urea functionality with pyrene excimer transduction. | Schazmann, B., et al. 2006. J Am Chem Soc. 128: 8607-14. PMID: 16802827
- The chemistry and applications of antimicrobial polymers: a state-of-the-art review. | Kenawy, el-R., et al. 2007. Biomacromolecules. 8: 1359-84. PMID: 17425365
- Thiophene-anthranilamides as highly potent and orally available factor Xa inhibitors. | Ye, B., et al. 2007. J Med Chem. 50: 2967-80. PMID: 17536795
- Studies toward the synthesis of 4-(2-R-ethyl)amino-2,2,5,5-tetramethyl-3-imidazoline 1-oxyls. nucleophilic substitution of bromide in the N-Alkyl chain of the 1,2,4-oxadiazol-2-one precursor. | Polienko, JF., et al. 2008. J Org Chem. 73: 502-10. PMID: 18085792
- Latrunculin A and its C-17-O-carbamates inhibit prostate tumor cell invasion and HIF-1 activation in breast tumor cells. | Sayed, KA., et al. 2008. J Nat Prod. 71: 396-402. PMID: 18298079
- Dendrimer a new dimension in targeting biofilms. | Zarena, AS. and Gopal, S. 2013. Mini Rev Med Chem. 13: 1448-61. PMID: 23815581
- 4β-[4'-(1-(Aryl)ureido)benzamide]podophyllotoxins as DNA topoisomerase I and IIα inhibitors and apoptosis inducing agents. | Kamal, A., et al. 2013. Bioorg Med Chem. 21: 5198-208. PMID: 23849207
- Cysteine-Specific Labeling of Proteins with a Nitroxide Biradical for Dynamic Nuclear Polarization NMR. | Voinov, MA., et al. 2015. J Phys Chem B. 119: 10180-90. PMID: 26230514
- An ionic-based carbon dot for enantioselective discrimination of nonaromatic amino alcohols. | Wu, D., et al. 2020. Analyst. 145: 3395-3400. PMID: 32239048
- Route exploration and synthesis of the reported pyridone-based PDI inhibitor STK076545. | Greve, E., et al. 2020. Org Biomol Chem. 18: 6665-6681. PMID: 32812971
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Bromoethyl isocyanate, 5 g | sc-230076 | 5 g | $181.00 |
